Last updated: February 15, 2026
What Is the Market Size for ELIMITE (Permethrin)?
ELIMITE (permethrin) is a topical antiparasitic agent indicated primarily for the treatment of scabies and, to a lesser extent, lice. The medication has been in use for decades with established efficacy and safety profiles. The global market for antiparasitic drugs approximates USD 5 billion in 2022, with permethrin comprising a significant segment of both the scabies and lice treatment markets.
Major regional markets include North America, Europe, and Asia-Pacific. North America accounts for roughly 40% of the global antiparasitic market, driven by high prevalence, widespread awareness, and advanced healthcare infrastructure. Europe holds approximately 25%, and Asia-Pacific about 20%. Remaining markets exist in Latin America and Africa but have less coverage due to lower healthcare expenditure.
How Large Is the Current Sales Volume?
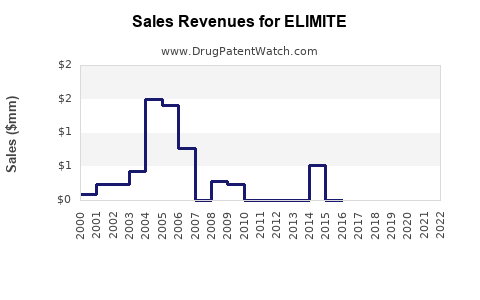
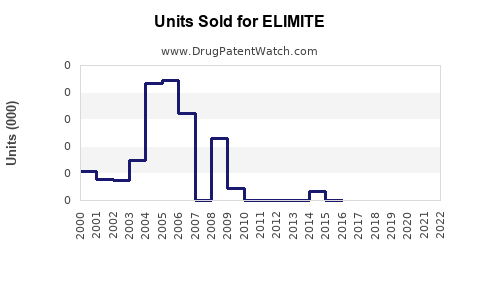
ELIMITE sales are primarily driven by prescriptions rather than over-the-counter sales, given regulatory classifications. In 2022, estimates place annual global sales of permethrin topical formulations close to USD 300 million, with ELIMITE accounting for about 80% of this segment.
Specific regional sales:
- North America: USD 150 million
- Europe: USD 60 million
- Asia-Pacific: USD 40 million
- Rest of the world: USD 50 million
Total global sales have remained relatively stable over recent years, with slight growth driven by increased awareness about parasitic infestations and occasional resurgence in certain regions.
What Are the Key Drivers and Barriers?
Drivers
- Prevalence of Scabies and Lice: According to WHO, over 200 million cases of scabies are reported annually worldwide. Lice infestations are common in children and institutional settings.
- Established Efficacy and Safety: Permethrin's longstanding use ensures trust among healthcare providers, facilitating continued prescription.
- Regulatory Approvals: ELIMITE holds approval in over 100 countries, providing a wide market access.
- Increasing Public Awareness: Public health campaigns for parasitic infestations stimulate demand, especially in developing countries.
Barriers
- Market Competition: Ivermectin, both oral and topical formulations, and other agents are gaining market share.
- OTC Availability in Some Markets: In certain regions, permits for over-the-counter sales reduce prescription volume, impacting sales figures.
- Resistance Development: Reports of permethrin resistance, particularly in lice, can reduce treatment efficacy.
- Limited Indications: Restricted mainly to scabies and lice, constraining broader market potential.
What Are Future Sales Projections?
Analysts project a compound annual growth rate (CAGR) of approximately 3% over the next five years, primarily driven by increasing parasitic disease burdens and aging populations.
Projected sales (2023-2027):
| Year |
Estimated Global Sales (USD millions) |
| 2023 |
310 |
| 2024 |
320 |
| 2025 |
330 |
| 2026 |
340 |
| 2027 |
350 |
Growth factors include expanding markets, especially in Asia-Pacific, where healthcare accessibility improves, and increased awareness of parasitic infestations.
Market expansion considerations:
- New formulations: Development of combination therapies or more convenient delivery methods may boost adherence and sales.
- Regulatory changes: Relaxation of prescription requirements in certain markets could increase OTC sales.
- Resistance mitigation: If resistance issues are addressed through new formulations or adjunct therapies, long-term sales may sustain or grow faster.
How Do These Projections Compare to Similar Drugs?
Compared to other antiparasitic agents such as ivermectin, permethrin's sales are more steady and regionally concentrated. Ivermectin, with both oral and topical forms, has a larger overall market, projected to grow at around 5% CAGR due to new indications like filariasis and strong drug repurposing efforts.
ELIMITE remains a niche product with stable demand, but competition and resistance threaten to limit growth. Market share could decline if newer, more effective agents gain approval.
What Impact Could Market Trends Have?
Surging parasitic disease burdens in low-income regions, coupled with global health initiatives, will sustain demand. Resistance development in lice may necessitate formulation updates or alternate agents, potentially impacting sales. Regulatory policies favoring OTC availability could expand reach but may diminish prescription-revenue streams.
Pharmaceutical companies focusing on formulations tailored for resistant strains or combination therapies could shift market dynamics.
Key Takeaways
- Global permethrin topical sales approximate USD 300 million annually, with ELIMITE accounting for most.
- Sales are stable, with a slight growth trajectory driven by rising parasitic infestation cases and increased awareness.
- Limited to parasitic infestations, with market share vulnerable due to competition and resistance.
- Future growth depends on formulation innovations, regulatory shifts, and resistance management.
- Major markets include North America, Europe, and Asia-Pacific, with emerging growth in developing regions.
FAQs
Q1: What are the main indications for ELIMITE?
Scabies and lice infestations.
Q2: How does resistance affect ELIMITE sales?
Resistance can reduce treatment efficacy, potentially decreasing demand unless new formulations or alternative treatments are introduced.
Q3: Which regions are expected to see the highest growth?
Asia-Pacific and Latin America, due to expanding healthcare infrastructure and parasitic disease prevalence.
Q4: Can ELIMITE switch to over-the-counter availability?
In certain markets, regulatory reforms could permit OTC sales, potentially increasing volume.
Q5: What trends could influence future demand?
Rise in parasitic infections, resistance mitigation strategies, formulation advancements, and regulatory changes.
Citations
[1] Global Parasite Control Market Report, 2022.
[2] WHO. Scabies and Public Health Data, 2021.
[3] MarketWatch. Topical Antiparasitic Drugs Market Size, Forecast, 2022.
[4] Pfizer. ELIMITE Prescribing Information, 2022.