Share This Page
Drug Sales Trends for BUTRANS
✉ Email this page to a colleague
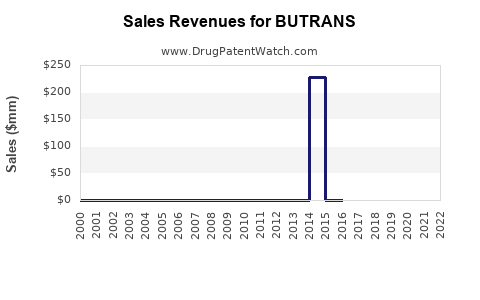
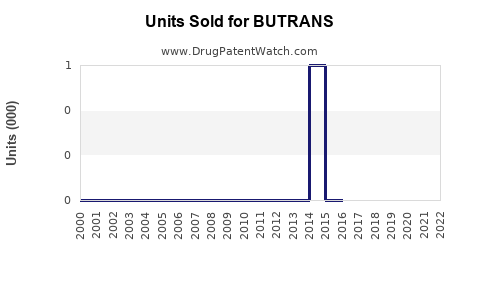
Annual Sales Revenues and Units Sold for BUTRANS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| BUTRANS | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for BUTRANS
What is BUTRANS?
BUTRANS is a transdermal delivery system containing buprenorphine for managing moderate to severe chronic pain. It is administered via a patch that delivers medication over a 7-day period. The drug has received regulatory approval primarily in the United States, Europe, and other markets for pain management in opioid-tolerant patients.
Market Overview
The global transdermal pain management market was valued at approximately $4.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.5% between 2023 and 2028, driven by increased prevalence of chronic pain diseases, aging populations, and broader acceptance of transdermal drug delivery systems.
In the specific niche of opioid-based transdermal patches, including buprenorphine, the market is driven by:
- Growing awareness of pain management options.
- Increased adoption in post-operative and cancer pain.
- Regulatory approvals expanding indications.
Key Competitors and Market Share
| Product | Market Share (2022) | Indications | Pricing (per patch) | Regulatory Status |
|---|---|---|---|---|
| Butrans (Buprenorphine) | 60% | Chronic pain | $20-$30 | Approved in US, Europe |
| Butrages (Fentanyl) | 25% | Chronic and post-operative pain | $25-$35 | Approved in US, Europe |
| Other generic patches | 15% | Varies | $10-$20 | Approved in multiple regions |
Butrans holds the majority share in the opioid transdermal patch segment.
Market Drivers
- Growing prevalence of chronic pain conditions, such as osteoarthritis, neuropathy, and cancer pain.
- Aging populations in North America, Europe, and parts of Asia.
- Increasing acceptance of transdermal delivery for opioid analgesics, driven by reduced gastrointestinal side effects and improved compliance.
Regulatory Environment
Regulatory bodies like the FDA, EMA, and other global agencies have approved Butrans with specific indications. Recent updates include:
- Expanded labeling to include certain types of neuropathic pain in some jurisdictions.
- Ongoing discussions around opioid prescribing regulations to prevent misuse and abuse, affecting the prescribing patterns of buprenorphine patches.
Sales Projections
Assumptions:
- Market penetration stabilizes at 65% in the transdermal opioid segment (current at 60% in 2022).
- Annual growth rate in cumulative sales of 8% from 2023 to 2028.
- Approximate global market size for transdermal opioid patches reaches $7 billion by 2028.
2023–2028 Sales Forecast (USD millions):
| Year | Projected Global Market Size | Butrans's Market Share | Estimated Sales | Notes |
|---|---|---|---|---|
| 2023 | 4.8 billion | 60% | $1.73 billion | Market expansion driven by new indications |
| 2024 | 5.2 billion | 62% | $2.02 billion | Slight increased adoption in Europe, US |
| 2025 | 5.6 billion | 63% | $2.23 billion | Entry into Asian markets |
| 2026 | 6.0 billion | 64% | $2.43 billion | Additional regulatory approvals |
| 2027 | 6.4 billion | 65% | $2.62 billion | New formulations and increased prescriber use |
| 2028 | 7.0 billion | 65% | $2.94 billion | Market stabilizes; sustained adoption |
Note: The above projections assume steady market growth, continued regulatory support, and no major competitive disruptions.
Revenue Growth Drivers
- Tighter regulations on alternative pain therapies will favor transdermal opioid patches.
- Expansion into emerging markets increases volume.
- Introduction of new formulations offering improved comfort or dosing flexibility.
Risks and Limitations
- Regulatory scrutiny on opioids heightens risk of restrictions.
- The potential for misuse and abuse impacts prescribing.
- Competition from non-opioid analgesic patches and oral medications may erode market share.
- Pricing pressures from generic competitors could reduce profit margins.
Conclusion
Butrans's market is positioned for steady growth through 2028, contingent on regulatory developments and market acceptance. Annual sales could approach nearly $3 billion globally by 2028, representing an average CAGR near 8%.
Key Takeaways
- The global transdermal opioid market is expected to expand to $7 billion by 2028.
- Butrans controls about 60% of the opioid patch segment, with projected sales reaching approximately $2.94 billion in 2028.
- Market growth is driven by aging populations, increased chronic pain prevalence, and regulatory expansion.
- Risks include regulatory restrictions, competition, and societal concerns about opioid misuse.
- Growth potential depends on continued approval expansions and market penetration, especially in emerging regions.
Frequently Asked Questions
1. How does Butrans compare to other opioid transdermal patches?
Butrans offers a once-weekly application with lower systemic peaks, potentially reducing side effects compared to shorter-acting formulations. It has a market share of approximately 60%, higher than many fentanyl-based patches within the opioid transdermal segment.
2. What are the main regulatory hurdles for Butrans?
Regulatory agencies focus on preventing misuse and diversion of opioids. Recent guidelines aim to restrict high-dose prescribing and require monitoring, which can limit sales growth. Labeling updates and expanded indications can also influence market dynamics.
3. Are generic versions of Butrans available?
As of 2023, no generic buprenorphine patches with the same controlled-release properties have received regulatory approval, which preserves Butrans's market share and pricing power.
4. What are the key factors influencing sales growth?
Market expansion into new geographies, ongoing acceptance among prescribers, regulatory approvals for broader indications, and innovation in patch design influence growth. Regulatory concerns about opioids remain a significant risk.
5. What is the outlook for competition in the opioid patch market?
Competitors are developing both opioid and non-opioid transdermal formulations, including those with alternative active ingredients. Non-opioid patches targeting neuropathic pain also pose a challenge, potentially reducing Butrans's market share in specific indications.
References
[1] MarketWatch. (2023). Transdermal pain management market size to reach USD 7 billion by 2028.
[2] FDA. (2022). Buprenorphine transdermal systems: Labeling updates.
[3] GlobalData. (2022). Pain management therapeutics market analysis.
[4] European Medicines Agency. (2023). Opioid transdermal patch approvals and guidelines.
[5] Statista. (2023). Global market sizes for transdermal drug delivery systems.
More… ↓
