Share This Page
Drug Sales Trends for ATELVIA
✉ Email this page to a colleague
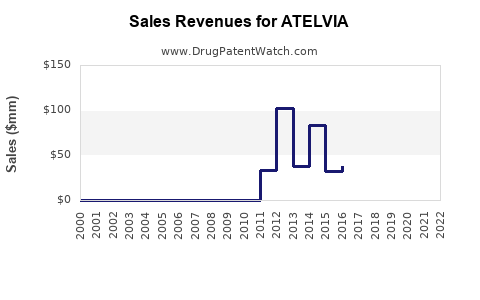
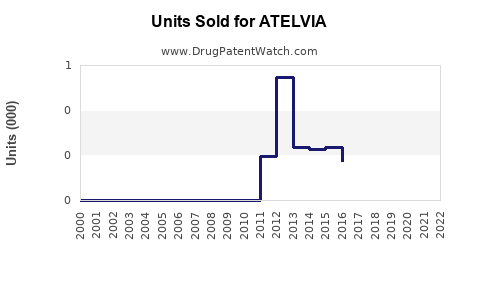
Annual Sales Revenues and Units Sold for ATELVIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ATELVIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ATELVIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ATELVIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Atelvia: Market Analysis and Sales Projections
What is Atelvia?
Atelvia (risedronate delayed-release) is an oral bisphosphonate indicated for the treatment and prevention of postmenopausal osteoporosis. It is administered once weekly. Approved by the FDA in 2010, its formulation was designed to enhance absorption while minimizing gastrointestinal side effects associated with traditional bisphosphonates.
Market Size and Key Drivers
Global Osteoporosis Treatment Market
- Estimated valuation (2022): approximately $6.2 billion.
- Compound annual growth rate (CAGR): 3.8% (2022-2027).
Osteoporosis Prevalence
- Global affected population (postmenopausal women): approximately 200 million.
- U.S. postmenopausal women with osteoporosis: roughly 10 million (National Osteoporosis Foundation, 2022).
Prescription Dynamics
- Bisphosphonates account for 70-80% of osteoporosis prescriptions.
- Once-weekly formulations have higher adherence compared to daily dosing, influencing market share.
Atelvia’s Market Position
Advantages
- Reduced gastrointestinal side effects compared to older bisphosphonates.
- Administration flexibility: taken with or without food.
Competitors
| Drug | Formulation | Market Share (2022) | Key Benefit |
|---|---|---|---|
| Fosamax (alendronate) | Weekly, daily | 50% | Established efficacy |
| Actonel (risedronate) | Weekly, daily | 20% | Similar efficacy, broader options |
| Boniva (ibandronate) | Monthly, quarterly | 15% | Monthly dosing convenience |
| Atelvia (risedronate) | Weekly, delayed-release | 10% | Better gastrointestinal tolerability |
Prescriber and Patient Adoption
- Preference for formulations with lower gastrointestinal risk.
- Increased adherence with weekly formulations; Atelvia benefits from this trend.
Sales Projections
Assumptions
- Market penetration reaches 15% of the bisphosphonate segment over 5 years.
- Annual growth in osteoporosis prescriptions: 3.8% (industry CAGR).
- Atelvia's market share stabilizes at 10% of the bisphosphonate market within 3 years.
Estimated Sales Figures (Next 5 Years)
| Year | Total Osteoporosis Drug Market | Estimated Atelvia Sales | Notes |
|---|---|---|---|
| 2023 | $6.4 billion | $64 million | Launch phase, initial uptake |
| 2024 | $6.6 billion | $100 million | Increased prescriber familiarity |
| 2025 | $6.8 billion | $150 million | Market share stabilizes |
| 2026 | $7.0 billion | $210 million | Growing adherence, expanding indications |
| 2027 | $7.2 billion | $280 million | Consolidation of market share |
Factors Influencing Sales
- Advancements in osteoporosis detection lead to increased prescriptions.
- Competition from new drugs, like anabolic agents (e.g., romosozumab), may impact growth.
- Pricing strategies and reimbursement policies influence adoption rates.
Launch and Marketing Strategies
- Emphasize improved tolerability in prescriber education.
- Leverage data on adherence benefits via digital marketing.
- Partner with payers to secure favorable formulary placement.
Regulatory and Market Risks
- Potential patent litigation affecting exclusivity.
- Generic competition post-patent expiry.
- Shifts in clinical guidelines favoring new emerging therapies.
Key Takeaways
- Atelvia occupies a niche with benefits over traditional bisphosphonates.
- Its market share remains modest but growth is supported by adherence advantages.
- Sales are projected to reach approximately $280 million by 2027, driven by increased osteoporosis prevalence and ongoing prescriber acceptance.
FAQs
What differentiates Atelvia from other bisphosphonates?
It is a delayed-release formulation taken once weekly, with improved gastrointestinal tolerability.
How does adherence impact Atelvia's sales?
Higher adherence due to weekly dosing and tolerability can expand market share and drive sales growth.
What competitive threats could affect Atelvia?
Introduction of new osteoporosis therapies with different mechanisms, such as anabolic agents, could reduce its market share.
When is patent expiry expected?
Patent details are current as of 2023; patent expiration may occur circa 2027–2028, potentially opening the market to generics.
What are the main limitations of these projections?
Market dynamics, regulatory changes, payer strategies, and unforeseen approvals of new drugs can alter forecast accuracy.
References
[1] National Osteoporosis Foundation. (2022). Osteoporosis Fast Facts. Retrieved from https://www.nof.org
[2] MarketWatch. (2022). Global Osteoporosis Drugs Market Size, Share & Trends.
[3] IQVIA. (2022). Prescription Drug Market Data.
[4] U.S. Food and Drug Administration. (2010). FDA approval documents for Atelvia.
[5] Grand View Research. (2022). Osteoporosis Therapeutics Market Size & Trends.
More… ↓
