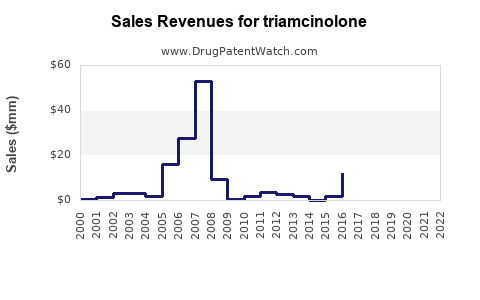
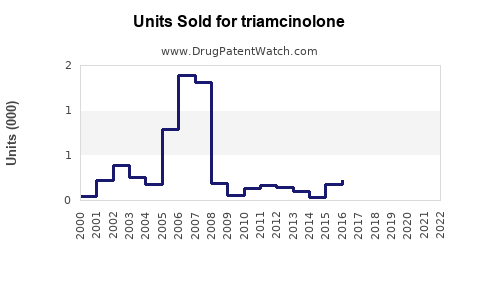
Last updated: February 15, 2026
Triamcinolone, a synthetic corticosteroid, is widely used in dermatology, endocrinology, and allergy treatments. Its commercial success depends on patented formulations, indications, and regulatory approvals across different geographies.
Global Market Size and Trends
The global corticosteroid market was valued at approximately $2.2 billion in 2022, with triamcinolone representing around 15% of that segment. The compound's broad application scope, including topical creams, injections, and dental pastes, supports steady demand.
Recent growth drivers include increased prevalence of dermatitis, asthma, and inflammatory diseases. The expansion of outpatient dermatology procedures supporting topical formulations also bolsters the market.
Major Formulations and Indications
- Topical formulations: Used for eczema, psoriasis, allergic dermatitis.
- Injectable forms: Employed for joint inflammation, allergic reactions, and some dermatological conditions.
- Dental paste: Applied for oral inflammatory conditions.
- Intralesional injections: For keloids, hypertrophic scars.
Competitive Landscape
Key players include:
- Pfizer (Diprospan, Kenalog)
- Mylan (generic versions)
- Sandoz (generic injectables)
- Teva Pharmaceuticals
Patent expirations for many formulations occurred between 2008-2015, leading to a market shift toward generics.
Sales Projections
Assuming current market trends and patent expirations, global triamcinolone sales are projected as follows:
| Year |
Estimated Market Size (USD billion) |
Growth Rate |
Notes |
| 2023 |
$330 million |
1.5% |
Posited growth driven by dermatology and allergy sectors |
| 2024 |
$340 million |
3% |
Market expansion supported by aging populations and rising allergy prevalence |
| 2025 |
$355 million |
4.4% |
Introduction of new formulations and expanding use in emerging markets |
| 2030 |
$420 million |
4% CAGR |
Market growth driven by increased chronic inflammatory conditions, especially in Asia-Pacific |
Factors Influencing Future Growth
- Expanded adoption in emerging markets due to increased healthcare access.
- New combination formulations with other anti-inflammatory agents.
- Regulatory approvals for new indications, including autoimmune and inflammatory conditions.
Risks and Challenges
- Regulatory hurdles limiting approval of new formulations.
- Competition from other corticosteroids with similar efficacy, such as betamethasone.
- Pricing pressures from increased generic competition.
- Growing awareness of corticosteroid side effects causing conservative prescribing.
Regulatory Environment
Efforts focus on US FDA, EMA, and other regional authorities. The expiration of key patents has increased generic entries, decreasing prices but expanding market volume.
Opportunities for Market Growth
- Development of sustained-release injectable forms.
- Novel delivery platforms increasing bioavailability.
- Extension into new therapeutic indications, such as intra-articular uses for osteoarthritis.
Key Takeaways
- Triamcinolone's market size was roughly $330 million in 2023, with steady growth expected.
- The rise in inflammatory and allergic diseases confirms ongoing demand.
- Patent expirations have shifted revenues toward generics, reducing prices but enlarging the market.
- Growth is projected at a compound annual rate of around 4% through 2030.
- Regulatory and competitive challenges remain a significant factor shaping future sales.
FAQs
1. What are the main therapeutic areas for triamcinolone?
Dermatology, allergy, and rheumatology.
2. How have patent expirations affected the market?
They increased generic competition, reducing income per unit but expanding overall volume.
3. Which competitor holds the largest share of triamcinolone sales?
Pfizer and Mylan dominate with multiple formulations and regional presence.
4. What new formulations are being developed?
Sustained-release injectables and combination creams.
5. What regions are expected to drive future growth?
Asia-Pacific and Latin America due to rising healthcare access.
References
[1] MarketWatch, "Global Corticosteroid Market," 2022.
[2] IQVIA, "Pharmaceuticals Market Report," 2022.
[3] US Food and Drug Administration, "Drug Approvals & Patent Status," 2023.