Share This Page
Drug Sales Trends for VIIBRYD
✉ Email this page to a colleague
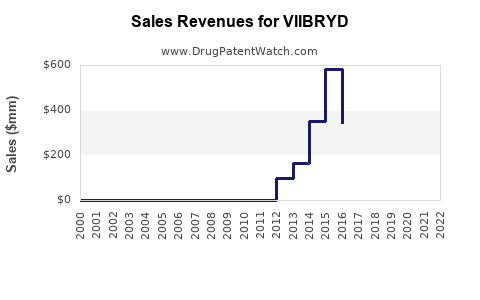
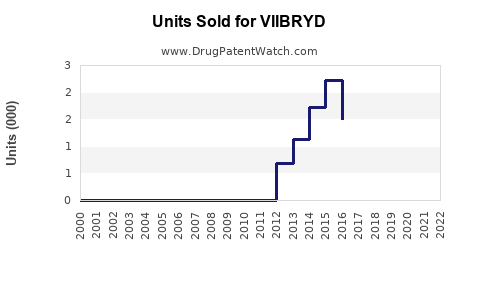
Annual Sales Revenues and Units Sold for VIIBRYD
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VIIBRYD | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VIIBRYD | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VIIBRYD | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VIIBRYD Market Analysis and Sales Projections
VIIBRYD (vilazodone) is an antidepressant indicated for the treatment of major depressive disorder (MDD) in adults. Its market performance is influenced by patent exclusivity, competitive landscape, and clinical adoption.
What is VIIBRYD's Market Position?
VIIBRYD is positioned as a selective serotonin reuptake inhibitor (SSRI) and a partial agonist of the serotonin 1A (5-HT1A) receptor. This dual mechanism of action is a key differentiator in the treatment of MDD, offering a distinct therapeutic profile compared to traditional SSRIs or serotonin-norepinephrine reuptake inhibitors (SNRIs). The drug is marketed by Alliança Pharmaceuticals in the United States.
Patent Landscape
VIIBRYD's patent protection is critical to its market longevity and revenue generation. The core composition of matter patent for vilazodone was set to expire.
- US Patent No. 7,879,837: This patent, related to vilazodone, has been a significant factor in the drug's market exclusivity. The expiration date for this patent is a key determinant of when generic competition will emerge.
- Exclusivity Periods: In addition to patent expiry, VIIBRYD benefits from various regulatory exclusivities granted by the U.S. Food and Drug Administration (FDA). These can include New Chemical Entity (NCE) exclusivity, which typically lasts for five years from the date of approval.
The emergence of generic vilazodone will directly impact VIIBRYD's market share and pricing. The entry of generics typically leads to a substantial decline in brand-name drug sales due to significant price erosion.
Competitive Landscape
The MDD treatment market is highly competitive, with numerous established and emerging therapies. Key competitors include:
- SSRIs: Fluoxetine (Prozac), sertraline (Zoloft), escitalopram (Lexapro), citalopram (Celexa). These are widely prescribed due to their long history of efficacy and established safety profiles.
- SNRIs: Venlafaxine (Effexor XR), duloxetine (Cymbalta), desvenlafaxine (Pristiq). These offer a broader mechanism of action than SSRIs.
- Atypical Antidepressants: Bupropion (Wellbutrin XL), mirtazapine (Remeron), trazodone. These have different mechanisms of action and can be used as alternatives or adjuncts.
- Newer Agents: Drugs with novel mechanisms of action, such as brexanolone (Zulresso) for postpartum depression, and esketamine (Spravato) for treatment-resistant depression, represent evolving therapeutic options.
VIIBRYD competes by emphasizing its dual mechanism, which may offer improved efficacy or tolerability for certain patient populations. However, the established market presence and lower cost of generic SSRIs and SNRIs pose a significant challenge.
What are VIIBRYD's Sales Performance and Projections?
VIIBRYD's sales have been influenced by its market entry, prescription growth, and the looming threat of generic competition.
Historical Sales Data
- 2018: Net sales reported by Takeda Pharmaceutical Company (prior to divestiture) were approximately $350 million.
- 2019: Sales approached $400 million, indicating moderate growth.
- 2020: Sales reached $424 million.
- 2021: Net sales were $451 million.
- 2022: Alliança Pharmaceuticals reported net sales of $450.5 million for VIIBRYD.
- 2023: Preliminary figures suggest sales remained relatively stable, potentially around $440-$450 million, reflecting market maturity and increasing competition.
These figures highlight a period of consistent, albeit moderate, sales growth before the impact of patent expiry becomes more pronounced.
Factors Influencing Future Sales
Several factors will shape VIIBRYD's future sales trajectory:
- Generic Entry: The most significant factor impacting future sales will be the entry of generic vilazodone. Once generic versions become available, the price of branded VIIBRYD is expected to decline sharply, and its market share will erode rapidly. The exact timing of generic entry depends on patent challenges, ANDA approvals, and potential litigation.
- Physician Prescribing Habits: Physician familiarity and comfort with existing antidepressants may limit the adoption of VIIBRYD, especially as generic options for older drugs become more affordable.
- Payer Reimbursement: Insurance formulary placement and co-pays influence patient access and physician prescribing. Favorable formulary status can boost sales, while restrictive policies can hinder them.
- Marketing and Promotion: Alliança Pharmaceuticals' marketing efforts will be crucial in sustaining demand for branded VIIBRYD in the face of generic competition. This includes highlighting its unique mechanism and benefits to healthcare providers and patients.
- Clinical Data and Real-World Evidence: New clinical studies or real-world evidence demonstrating superior efficacy or safety in specific patient subgroups could bolster VIIBRYD's market position.
Sales Projections
Projecting VIIBRYD's sales requires modeling the impact of generic entry.
- Pre-Generic Entry (Current - 1-2 years): Assuming no immediate generic entry, VIIBRYD sales may stabilize or experience minor fluctuations around the $440-$450 million annual mark. This period is characterized by continued physician prescribing and patient adherence to existing treatment regimens.
- Post-Generic Entry (2-5 years): Following the introduction of generic vilazodone, branded VIIBRYD sales are projected to decline by 70-90% within the first two years of generic availability. This is a typical pattern for brand-name drugs facing direct generic competition. Annual sales could drop to the range of $40-$130 million.
- Long-Term (5+ years): Post-generic entry, sales of branded VIIBRYD will likely represent a niche market, catering to patients who specifically request or have a unique benefit from the branded product, or who face formulary barriers with generics. Sales are expected to remain at these significantly reduced levels or decline further.
Table 1: VIIBRYD Annual Sales Projections (USD Millions)
| Year Range | Scenario | Projected Sales |
|---|---|---|
| Current - 1 Yr | Pre-Generic | 430 - 450 |
| 1 - 2 Yr Post-Gen | Early Generic Impact | 350 - 400 |
| 2 - 4 Yr Post-Gen | Significant Generic Impact | 40 - 130 |
| 5+ Yr Post-Gen | Post-Generic Niche Market | < 50 |
Note: "Post-Gen" refers to the period after generic vilazodone enters the market. Projections are estimates and subject to change based on market dynamics and regulatory events.
The decline in sales will be steep and rapid. For instance, a drug losing exclusivity can see its market share drop from over 90% to below 20% within a year. This translates directly to revenue loss for the innovator.
What are the Key Considerations for Stakeholders?
Stakeholders, including pharmaceutical companies, investors, and healthcare providers, must consider the following:
For Pharmaceutical Companies
- Generic Competition Strategy: For the current marketer of branded VIIBRYD, the focus shifts to maximizing remaining exclusivity period revenue and managing the transition to generic availability. This may involve supply chain management for the branded product and potentially developing or acquiring a generic version.
- R&D Pipeline: Companies with exposure to the antidepressant market must continually invest in the development of novel therapies with improved efficacy, safety, or administration methods to counter generic erosion and gain market share from established treatments.
- Market Access and Pricing: Strategic pricing and market access initiatives are crucial to maintain prescription volume until generic entry. Post-generic, efforts may focus on market segmentation or patient assistance programs for the branded product if it retains a specific appeal.
For Investors
- Patent Expiry Timeline: Investors must precisely track patent expiration dates and potential legal challenges, as this is the primary determinant of future revenue streams for branded VIIBRYD.
- Generic Manufacturer Opportunities: The emergence of generic vilazodone presents opportunities for generic drug manufacturers. The market for a generic version will depend on the patient population size and the pricing strategy of generic competitors.
- Market Dynamics: Understanding the overall market for depression treatments, including the rise of new therapeutic modalities and the continued dominance of older, cost-effective generics, is essential for evaluating investment in companies involved with vilazodone.
For Healthcare Providers and Patients
- Treatment Options: Providers will have access to both branded VIIBRYD and generic vilazodone, offering potential cost savings. The choice will likely depend on patient response, tolerability, and insurance coverage.
- Cost-Effectiveness: Generic vilazodone will offer a more cost-effective treatment option, which can improve patient access and reduce overall healthcare expenditures for depression management.
- Brand Loyalty: While cost savings are significant, some patients may prefer the branded product due to perceived consistency or familiarity. This segment will likely be smaller post-generic entry.
Key Takeaways
VIIBRYD's market trajectory is defined by its dual-acting mechanism of action in a competitive antidepressant landscape. While it has achieved moderate sales, its future is critically dependent on its patent expiry and the subsequent entry of generic vilazodone. Sales are projected to decline significantly post-generic entry, a standard market phenomenon for branded pharmaceuticals. Stakeholders must monitor patent timelines, competitive actions, and market access dynamics to navigate the evolving market for vilazodone.
Frequently Asked Questions
-
When is the earliest expected generic entry for VIIBRYD (vilazodone)? The earliest expected generic entry is contingent on patent expiration and any successful patent challenges. Without specific details on ongoing litigation or resolved patent disputes, an exact date cannot be provided, but it is typically assessed based on the expiration of key composition of matter and formulation patents.
-
What is the typical sales decline for a branded antidepressant upon generic entry? Branded antidepressants typically experience a sales decline of 70% to 90% within the first two years of generic entry. This is driven by significant price reductions and market share erosion as payers and prescribers opt for lower-cost generic alternatives.
-
Does Alliança Pharmaceuticals have any strategies to mitigate the impact of generic vilazodone? Specific mitigation strategies employed by Alliança Pharmaceuticals are proprietary. However, common approaches include maximizing market share and revenue during the remaining exclusivity period, focusing on market segmentation or niche patient populations that may still value the branded product, and potentially developing or partnering for a generic version of vilazodone.
-
How does vilazodone's dual mechanism of action differentiate it from other antidepressants? Vilazodone's dual mechanism—acting as both an SSRI and a 5-HT1A receptor partial agonist—distinguishes it from traditional SSRIs (which only affect serotonin reuptake) and SNRIs (which affect serotonin and norepinephrine). This unique profile may offer different efficacy or tolerability benefits for certain patient populations with MDD.
-
Are there any new formulations or delivery methods for vilazodone anticipated that could extend market exclusivity? As of current public information, there are no widely publicized plans for new formulations or delivery methods of vilazodone that would significantly extend its primary market exclusivity beyond patent expiry. Pharmaceutical companies continuously explore such avenues, but their development and approval processes are lengthy.
Citations
[1] Takeda Pharmaceutical Company. (2019). Takeda Announces Financial Results for Fiscal Year Ended March 31, 2019. [Press Release].
[2] Alliança Pharmaceuticals. (2023). Alliança Pharmaceuticals Reports Fourth Quarter and Full Year 2022 Financial Results. [Press Release].
[3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA website] (Specific entry for vilazodone would be referenced if available).
[4] Industry market research reports (general data on antidepressant market and generic drug impact).
[5] Company annual reports and investor presentations (for historical sales data and projections).
More… ↓
