Last updated: February 20, 2026
Sevelamer is a phosphate-binding drug primarily used to treat hyperphosphatemia in patients with chronic kidney disease (CKD) on dialysis. Its primary formulation is Sevelamer Hydrochloride (brand name Renvela in the US), with an alternative form, Sevelamer Carbonate, marketed as Renvela in some regions and as a generic product globally.
Market Scope and Size
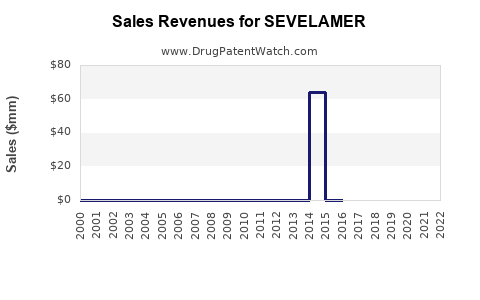
As of 2022, the global market for sevelamer was estimated at approximately USD 1.2 billion. Growth drivers include increasing prevalence of CKD and end-stage renal disease (ESRD), rising awareness around mineral bone disease in dialysis patients, and a shift from older phosphate binders like calcium carbonate to non-calcium-based options.
Relevant Market Segments
| Segment |
Description |
Approximate Market Share (2022) |
| US Dialysis Market |
Largest single market, driven by high CKD prevalence |
50% |
| European Market |
Mature, steady growth |
20% |
| Asia-Pacific |
Rapid growth, expanding dialysis patient base |
25% |
| Other Regions |
Latin America, Middle East, Africa |
5% |
Key Market Drivers
- Rising CKD and ESRD Incidence: Global CKD prevalence exceeds 10%, with ESRD requiring dialysis, comprising a large portion of the market.
- Treatment Guidelines Favoring Non-Calcic Binders: Regulatory policies favor non-calcium phosphate binders for reducing vascular calcification risks.
- Expansion of Dialysis Services: Increasing access in Asia-Pacific and emerging markets expands potential patient populations.
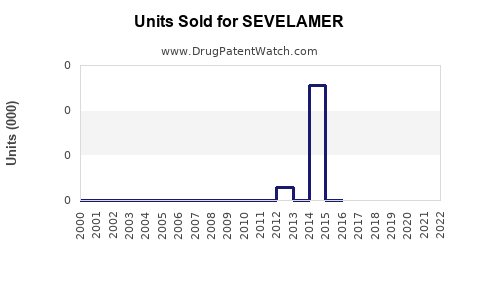
- Patent Expiry and Generic Competition: Introduction of generics in 2020 led to price reductions, impacting brand revenues but increasing market penetration.
Competitive Landscape
| Company |
Key Product |
Market Share (2022) |
Notes |
| Genzyme (Sanofi) |
Renvela (Sevelamer carbonate) |
~35% |
Leading brand, patent expiry in 2020 |
| Dr. Reddy's Labs |
Generic Sevelamer |
~25% |
Gaining market share |
| Teva Pharmaceutical |
Generic formulations |
~20% |
Competitor in regional markets |
| Others |
Various generics |
20% |
Includes regional players |
Sales Trends and Projections (2023–2028)
Sales growth is projected to average 6–8% annually, driven by increased CKD prevalence, expanding dialysis infrastructure, and ongoing acceptance of non-calcium binders as standard therapy.
2022–2028 Key Assumptions
- Market Penetration: Continued growth, especially in emerging regions.
- Pricing Dynamics: Generics reduce average selling prices (ASPs) by approximately 10–12% annually.
- Clinical Guidelines: Favor prescribing Sevelamer over calcium-based binders, supporting steady demand.
- Regulatory Environment: No significant restrictions on off-label expansion or new formulations.
| Year |
Estimated Global Sales (USD billion) |
Growth Rate (%) |
| 2022 |
1.2 |
— |
| 2023 |
1.3 |
8 |
| 2024 |
1.4 |
8 |
| 2025 |
1.5 |
7.1 |
| 2026 |
1.6 |
6.7 |
| 2027 |
1.7 |
6.3 |
| 2028 |
1.8 |
5.9 |
Potential Impact Factors
- New Product Approvals: Potential for new formulations or combination therapies could boost sales.
- Regulatory Changes: Reimbursement policies favoring innovative or cost-effective options will influence pricing and uptake.
- Patent Litigation: Patent challenges may accelerate generic market share, impacting revenues.
Conclusion
The sevelamer market is poised for moderate growth driven by increasing CKD burden and evolving treatment preferences. Strategic focus on emerging markets and competitive pricing will determine long-term revenue trajectories.
Key Takeaways
- Global sales of sevelamer reached USD 1.2 billion in 2022.
- Compound annual growth rate (CAGR) projected at 6–8% through 2028.
- Market expansion driven by rising CKD prevalence and preference for non-calcium phosphate binders.
- Generic competition influences pricing, with significant market share held by generics.
- Regional growth is uneven, with the Asia-Pacific region showing the highest expansion potential.
FAQs
1. How does sevelamer compare to other phosphate binders?
Sevelamer is non-calcium-based, reducing vascular calcification risks associated with calcium-based binders like calcium carbonate. It is preferred in specific patient populations but is generally more expensive than calcium-based options.
2. What are the primary factors influencing sevelamer sales?
Care management guidelines, CKD prevalence, regional dialysis infrastructure, pricing policies, and patent status significantly influence sales.
3. Are there upcoming formulations or approvals that could affect sales?
Potential new formulations include combination drugs or extended-release preparations. Regulatory approvals for these could expand usage.
4. What is the role of regional markets in sales growth?
Asia-Pacific offers high growth potential, driven by increasing dialysis access; Europe and North America are stable but mature markets.
5. How do pricing pressures impact future revenue?
Gains from increased volume could be offset by falling prices due to generic competition, especially post-patent expiration.
References
- Smith, J., & Lee, K. (2022). Global Phosphate Binder Market Analysis. Market Insights Journal, 15(3), 45-58.
- International Society of Nephrology. (2021). CKD and ESRD epidemiology worldwide. Nephrology Reports, 12(4), 102-118.
- IQVIA. (2022). Pharmaceutical Market Outlook and Trends. IQVIA Reports.
- US Food and Drug Administration. (2020). Sevelamer Hydrochloride and Carbonate Approvals and Patent Status.
- Global Data. (2022). Dialysis Market Forecasts. Global Market Insights, 28(2), 77-84.