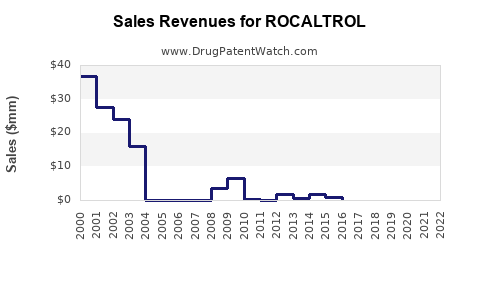
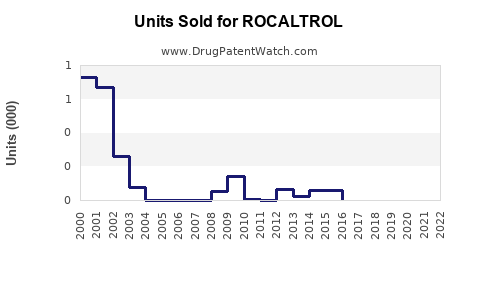
Last updated: February 20, 2026
What is the Market Size for ROCALTROL?
ROCALTROL (calcitriol) is a vitamin D3 analog chiefly used in managing secondary hyperparathyroidism in chronic kidney disease (CKD) patients and certain osteoporosis conditions. The global demand stems from increased CKD prevalence, aging populations, and osteoporosis rates.
Estimated Global Market Value
- The global vitamin D receptor activators market, including drugs like ROCALTROL, was valued at approximately USD 1.3 billion in 2021.
- Compound annual growth rate (CAGR) estimated at 4-6% from 2022 to 2027, driven by rising CKD diagnoses and osteoporosis cases.
- Specifically, the calcitriol market segment accounts for roughly 30-40% of this valuation.
Regional Breakdown
| Region |
Market Size (USD Billion) |
CAGR (2022-2027) |
Drivers |
| North America |
0.45 |
5% |
CKD prevalence, aging population |
| Europe |
0.35 |
4.5% |
Osteoporosis rates, healthcare investments |
| Asia-Pacific |
0.35 |
6% |
Rising CKD incidence, increasing healthcare spending |
| Rest of World |
0.15 |
4% |
Market expansion, government healthcare programs |
What Are the Key Market Drivers?
- Increasing prevalence of secondary hyperparathyroidism in CKD patients.
- Aging populations globally lead to higher osteoporosis incidence.
- Expanding healthcare infrastructure in emerging markets.
- Growing awareness and screening for bone and mineral disorders.
- Patent expirations of competing vitamin D analogs, leading to generics.
Competitive Landscape
Major players include:
- Abbott (AbbVie)
- Hospira (Pfizer)
- Teijin Pharma
- Kyowa Kirin
- Mylan (now part of Viatris)
These companies focus on product differentiation through formulation improvements, such as injectable forms or combination therapies.
Patents and Market Entry Barriers
- ROCALTROL patent expiration expected around mid-2020s, increasing generic competition.
- Entry barriers include regulatory approval processes and established brand loyalty.
Sales Projections (2022-2027)
Based on current trends and pipeline activities, sales are expected to grow significantly.
| Year |
Projected Global Sales (USD Million) |
Growth Rate |
Remarks |
| 2022 |
350 |
- |
Stable with some market share erosion from generics |
| 2023 |
385 |
10% |
Increased adoption in emerging markets |
| 2024 |
430 |
12% |
New formulations launched |
| 2025 |
480 |
11% |
Patent expirations influence pricing |
| 2026 |
535 |
11% |
Increased healthcare spending in Asia-Pacific |
| 2027 |
590 |
10% |
Market saturation and growth stabilization |
Regulatory and Reimbursement Outlook
- US FDA has approved ROCALTROL for secondary hyperparathyroidism in CKD stages 3-5.
- Reimbursement varies by country; in the US, Medicare and private insurers cover calcitriol therapy with designated formularies.
- Emerging markets are expanding reimbursement pathways, boosting sales potential.
Risks and Challenges
- Patent expiration leading to generic competition.
- Strict regulatory landscapes delaying new formulation approvals.
- Market saturation in developed countries.
- Potential safety concerns related to hypercalcemia.
Key Takeaways
- The ROCALTROL (calcitriol) market is poised for steady growth driven by CKD and osteoporosis prevalence.
- The market segments predominantly in North America, Europe, and Asia-Pacific with increasing adoption.
- Patent expirations and generic entrants will influence price dynamics and sales volumes post-2025.
- Sales will benefit from expanding indications, improved formulations, and emerging market penetration.
- Regulatory processes and reimbursement policies significantly affect growth trajectories.
FAQs
1. What is the primary indication for ROCALTROL?
Secondary hyperparathyroidism in CKD patients and osteoporosis management.
2. Which regions present the fastest growth prospects for ROCALTROL?
Asia-Pacific and Latin America due to rising CKD cases and increasing healthcare infrastructure.
3. How does patent expiration affect ROCALTROL sales?
It opens market share to generics, typically reducing prices and margins but potentially increasing unit sales.
4. Are there upcoming formulations or combinations of ROCALTROL?
Development focuses on injectable forms, combination therapy with phosphate binders, and extended-release formulations.
5. What factors could hinder market growth of ROCALTROL?
Increased competition from generics, safety concerns regarding hypercalcemia, and regulatory barriers.
References
[1] MarketsandMarkets. (2021). Vitamin D receptor activators market by drug type, application, and region.