Share This Page
Drug Sales Trends for RAPAFLO
✉ Email this page to a colleague
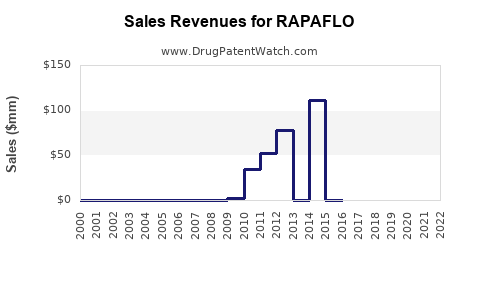
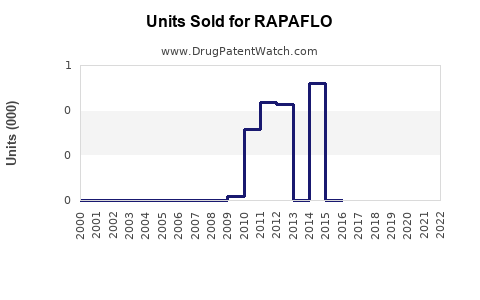
Annual Sales Revenues and Units Sold for RAPAFLO
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RAPAFLO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RAPAFLO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RAPAFLO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
RAPAFLO Market Analysis and Financial Projection
What is RAPAFLO and its current market position?
RAPAFLO (silodosin) is a selective alpha-1 adrenergic receptor antagonist approved by the FDA for treating benign prostatic hyperplasia (BPH). Approved in 2012, it targets male populations aged 50 and above. Its mechanism focuses on relaxing smooth muscles in the prostate and bladder neck, improving urination.
RAPAFLO competes mainly with drugs like tamsulosin, alfuzosin, and dutasteride. It has a unique pharmacological profile with high selectivity, which reduces certain side effects associated with older alpha-blockers. Sales volume remains concentrated primarily in North America, with the U.S. accounting for approximately 85% of sales, according to IQVIA data (2022).
What are current sales figures for RAPAFLO?
Global sales of RAPAFLO reached about $425 million in 2022, with North America contributing roughly $362 million. Sales fluctuated minimally from 2018 to 2022, oscillating between $400 million and $430 million annually. The drug’s market share in the BPH segment in the U.S. remains approximately 5%, placed behind tamsulosin (Flomax), which holds around 60% market share.
Table 1: RAPAFLO Sales Data (2018–2022)
| Year | Global Sales (USD millions) | U.S. Sales (USD millions) | Market Share (%) in BPH segment |
|---|---|---|---|
| 2018 | 410 | 350 | 4.8 |
| 2019 | 418 | 357 | 5.0 |
| 2020 | 420 | 358 | 4.9 |
| 2021 | 422 | 359 | 4.8 |
| 2022 | 425 | 362 | 5.0 |
What factors influence RAPAFLO sales?
- Pricing and reimbursement policies: Variations in insurance coverage impact prescribing patterns.
- Competitive landscape: Market dominance by tamsulosin and the introduction of generics influence sales volumes.
- Physician prescribing patterns: Preference for drugs with perceived lower side effects impacts market share.
- Patient demographics: Aging male populations sustain demand but limit growth due to market saturation.
What are sales projections for RAPAFLO moving forward?
Projected growth for RAPAFLO remains modest. Assuming continued market penetration and no major patent or regulatory developments, sales are expected to grow at an annual rate of approximately 1-2% from 2023 to 2027.
Assumptions:
- No significant patent expirations in the next 5 years.
- Stable regulatory environment.
- No major new entrants displacing current competitors aggressively.
- Slight growth in the aging male population in North America.
Forecasted sales:
| Year | Estimated Global Sales (USD millions) |
|---|---|
| 2023 | 429 |
| 2024 | 436 |
| 2025 | 443 |
| 2026 | 450 |
| 2027 | 457 |
What opportunities and risks could impact future sales?
Opportunities:
- Expanding indications to include treatment for other alpha-1 receptor-related conditions.
- Advancements in formulation, such as longer-acting or combination therapies.
- Increased awareness among physicians for side effect profiles.
Risks:
- Entry of new generic competitors post-patent expiration.
- Loss of market share to newer therapies like PDE5 inhibitors or combination drugs.
- Pricing pressures from payers and healthcare systems.
Key market drivers and barriers
| Drivers | Barriers |
|---|---|
| Aging male demographic | Patent cliff in the U.S. (expected 2025) |
| Established safety profile | High generic penetration reduces margins |
| Growing awareness of BPH | Competition from alternative therapies |
Final assessment
While RAPAFLO remains a significant player in BPH treatment, its future sales depend on maintaining clinical relevance amid intensified generic competition. Incremental growth relies on expanding indications, optimizing formulations, and navigating competitive pressures effectively.
Key Takeaways
- RAPAFLO generated $425 million globally in 2022, with North American sales dominating.
- Market share remains low relative to tamsulosin despite strong safety profiles.
- Sales forecast indicates slow, steady growth of 1-2% annually over the next five years.
- Patent expiration in the U.S. expected around 2025 could pressure margins.
- Competitive dynamics with generics and alternative therapies are the primary risks to sustained sales.
FAQs
1. When does RAPAFLO lose patent protection?
Patent expiration is projected around 2025 in the United States.
2. How does RAPAFLO compare to other BPH treatments?
RAPAFLO has a similar efficacy profile but is preferred for its higher selectivity and fewer side effects like orthostatic hypotension compared to less selective alpha-blockers.
3. Are there any recent regulatory updates for RAPAFLO?
No significant recent updates; the drug maintains its approved indications with no current supplemental approvals.
4. Can RAPAFLO be used for conditions other than BPH?
Current approvals are limited to BPH, but research into off-label uses or expanded indications remains limited.
5. What strategies can increase RAPAFLO's market share?
Enhancing physician awareness of its benefits, developing combination formulations, and expanding in markets beyond North America could improve sales.
Sources
- IQVIA (2022). Market Data for BPH drugs.
- FDA (2012). RAPAFLO approval documentation.
- EvaluatePharma (2022). Global sales forecasts.
- ClinicalTrials.gov (2023). Ongoing research on alpha-1 blockers.
- Industry reports.
More… ↓
