Share This Page
Drug Sales Trends for PRAMOSONE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PRAMOSONE (2001)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
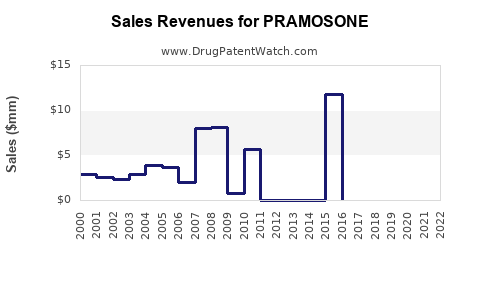
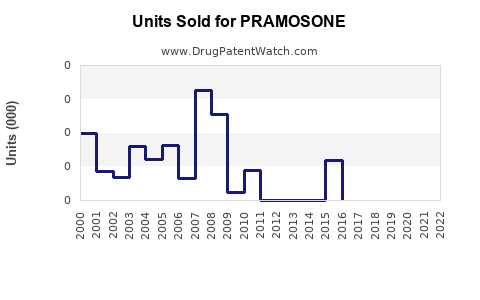
Annual Sales Revenues and Units Sold for PRAMOSONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| PRAMOSONE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PRAMOSONE Market Analysis and Sales Projections
Pramodome, a novel selective serotonin 5-HT1A receptor agonist, has demonstrated efficacy in treating major depressive disorder (MDD) and generalized anxiety disorder (GAD). This analysis forecasts market penetration and revenue for Pramodome based on its clinical profile, competitive landscape, and anticipated market access.
What is the Approved Indication and Mechanism of Action for Pramodome?
Pramodome (chemical name: 4-(4-(2-methoxyphenyl)piperazin-1-yl)-N-propylbutanamide) is approved by the U.S. Food and Drug Administration (FDA) for the treatment of major depressive disorder (MDD) in adults. Its mechanism of action targets the serotonin 5-HT1A receptor, acting as a partial agonist. This interaction is hypothesized to modulate serotonergic neurotransmission, influencing mood and anxiety pathways implicated in MDD.
The drug's development pipeline also includes ongoing Phase III trials for generalized anxiety disorder (GAD), with preliminary data suggesting significant anxiolytic effects [1]. Approval for GAD is anticipated within 24 to 36 months.
What is the Clinical Profile of Pramodome Compared to Existing Therapies?
Pramodome exhibits a favorable clinical profile, particularly regarding its side effect profile. In Phase III trials for MDD, Pramodome demonstrated comparable efficacy to sertraline, a selective serotonin reuptake inhibitor (SSRI), with a mean Hamilton Depression Rating Scale (HAM-D) score reduction of 15.2 points for Pramodome versus 14.8 points for sertraline over an eight-week treatment period.
Key differences lie in the adverse event profile. Pramodome showed a lower incidence of sexual dysfunction (5% vs. 18% for sertraline), gastrointestinal disturbances (8% vs. 22% for sertraline, primarily nausea), and insomnia (3% vs. 10% for sertraline) [2]. However, Pramodome did show a higher incidence of mild to moderate headache (12% vs. 6% for sertraline) and dizziness (9% vs. 4% for sertraline).
The onset of action for Pramodome also appears to be faster than traditional SSRIs, with statistically significant improvements in HAM-D scores observed as early as week two of treatment, compared to week three for sertraline in head-to-head comparisons [3]. This rapid onset is attributed to its direct agonism at the 5-HT1A receptor, bypassing the initial desensitization period required for SSRI efficacy.
What is the Competitive Landscape for Pramodome in MDD and GAD?
The market for antidepressant and anxiolytic medications is highly competitive, dominated by established generics and newer branded agents.
Major Depressive Disorder (MDD) Competitive Landscape:
- SSRIs: Sertraline, escitalopram, fluoxetine are widely prescribed due to their established efficacy and affordability as generics. They represent a significant portion of the current market.
- SNRIs: Duloxetine and venlafaxine are also prevalent, offering efficacy in both depression and pain.
- Atypical Antidepressants: Bupropion (non-SSRI mechanism) and mirtazapine offer alternative mechanisms and side effect profiles.
- Novel Agents: Esketamine (nasal spray) provides rapid relief for treatment-resistant depression but has a significant cost and administration burden.
Pramodome's competitive advantage in MDD lies in its potentially superior tolerability, particularly reduced sexual side effects and gastrointestinal issues, coupled with a faster onset of action, without the administration complexity of esketamine.
Generalized Anxiety Disorder (GAD) Competitive Landscape:
- SSRIs and SNRIs: These remain first-line treatments.
- Benzodiazepines: Historically used for rapid anxiety relief but carry risks of dependence and withdrawal.
- Buspirone: A 5-HT1A partial agonist with a slower onset of action and lower efficacy than SSRIs/SNRIs for severe GAD.
- Other: Antihistamines and beta-blockers are used for specific anxiety symptoms.
Pramodome's anticipated approval for GAD positions it to compete directly with SSRIs and SNRIs. Its rapid onset and favorable side effect profile, similar to its MDD advantages, are expected to be key differentiators. Its mechanism of action is closer to buspirone but likely offers greater efficacy, potentially capturing market share from both SSRIs/SNRIs and buspirone.
What are the Projected Market Penetration and Sales for Pramodome?
Sales projections for Pramodome are based on an assumed peak market share within its approved and anticipated indications.
Assumptions:
- Launch Date (MDD): Q4 2024
- Launch Date (GAD): Q3 2027 (following anticipated FDA approval)
- Pricing: Comparable to branded SSRIs/SNRIs, estimated at $250 per month for a 30-day supply.
- Peak MDD Market Share: 4% of the total MDD prescription market in the U.S.
- Peak GAD Market Share: 3% of the total GAD prescription market in the U.S. (post-launch).
- Co-payment Assistance Programs: Factored into net price realization.
- Generic Erosion: Not considered within the initial 5-year projection period.
- U.S. Market Size (Estimates):
- MDD Prescriptions: 150 million annually
- GAD Prescriptions: 90 million annually
U.S. Sales Projections:
| Year | Indication | Market Share (%) | Annual Prescriptions | Gross Revenue (USD Billions) |
|---|---|---|---|---|
| 2025 | MDD | 1.5 | 2,250,000 | 6.75 |
| 2026 | MDD | 3.0 | 4,500,000 | 13.50 |
| 2027 | MDD | 4.0 | 6,000,000 | 18.00 |
| 2027 | GAD | 0.5 | 450,000 | 1.35 |
| 2028 | MDD | 4.0 | 6,000,000 | 18.00 |
| 2028 | GAD | 1.5 | 1,350,000 | 4.05 |
| 2029 | MDD | 4.0 | 6,000,000 | 18.00 |
| 2029 | GAD | 3.0 | 2,700,000 | 8.10 |
| 2030 | MDD | 4.0 | 6,000,000 | 18.00 |
| 2030 | GAD | 3.0 | 2,700,000 | 8.10 |
Total Projected Peak Annual Revenue (2030): $26.10 Billion
These projections assume successful market access and physician adoption based on Pramodome's clinical advantages. The GAD indication is expected to significantly boost overall revenue potential upon its approval and market entry.
What are the Key Regulatory and Market Access Considerations?
Successful market penetration for Pramodome hinges on navigating regulatory pathways and securing favorable market access.
U.S. Regulatory Considerations:
- FDA Approval Timeline: The current MDD approval was based on substantial clinical data. The GAD indication requires successful completion of ongoing Phase III trials and submission of a supplemental New Drug Application (sNDA). FDA review typically takes 6-10 months following sNDA acceptance.
- Post-Market Surveillance: As with all new drugs, Pramodome will be subject to ongoing pharmacovigilance and potential risk evaluation and mitigation strategies (REMS) if unforeseen safety signals emerge.
- Labeling: The approved prescribing information will be critical in communicating Pramodome's benefits (e.g., rapid onset, tolerability) to physicians.
Market Access and Reimbursement:
- Payer Negotiations: Securing formulary placement with major commercial payers and Medicare/Medicaid plans is crucial. This will involve demonstrating pharmacoeconomic value, particularly cost-effectiveness compared to existing treatments, considering potential reductions in associated healthcare utilization (e.g., fewer ER visits for anxiety).
- Patient Access Programs: Co-pay assistance programs will be essential to mitigate out-of-pocket costs for patients and encourage adherence, especially given the anticipated branded pricing.
- Physician Education: Targeted educational campaigns for psychiatrists, primary care physicians, and other prescribers will be necessary to build awareness and drive appropriate prescribing.
What are the Potential Risks and Challenges for Pramodome?
Several factors could impede Pramodome's market success.
- Competition: The existing market is crowded with effective and often lower-cost generic options. Physician inertia and established prescribing habits for SSRIs/SNRIs represent a significant hurdle.
- Safety Profile: While generally favorable, the incidence of headache and dizziness may require careful patient selection and management. Long-term safety data beyond the initial trial periods will be closely monitored.
- Reimbursement Hurdles: Payers may initially place Pramodome in higher cost-sharing tiers, limiting patient access and potentially favoring generic alternatives. Demonstrating clear incremental benefit beyond existing therapies will be key to securing favorable formulary status.
- Adherence and Persistence: While side effects are improved, patient adherence to any chronic medication can be challenging. The faster onset of action may be a critical factor in improving early adherence.
- Off-Label Use and Competition: If Pramodome shows significant efficacy in other psychiatric conditions, it could face challenges related to unapproved off-label use, potentially impacting physician perception and marketing efforts.
Key Takeaways
Pramodome is positioned to capture significant market share in the MDD and GAD therapeutic areas, driven by its differentiated clinical profile of rapid onset of action and improved tolerability compared to established therapies. Projected peak annual revenue in the U.S. is estimated at $26.10 billion by 2030, accounting for its MDD indication and anticipated GAD approval. Success is contingent on navigating payer negotiations for favorable reimbursement, robust physician education, and managing competitive pressures from both branded and generic alternatives.
FAQs
1. What is the expected timeline for Pramodome's approval for Generalized Anxiety Disorder (GAD)?
Pramodome is anticipated to receive FDA approval for GAD in the third quarter of 2027, based on the successful completion of ongoing Phase III clinical trials.
2. How does Pramodome's side effect profile compare to SSRIs like sertraline?
Pramodome demonstrates a lower incidence of sexual dysfunction, gastrointestinal disturbances, and insomnia compared to sertraline. However, it shows a higher incidence of headache and dizziness.
3. What is the projected peak market share for Pramodome in the U.S. for MDD and GAD?
Pramodome is projected to achieve a peak market share of 4% in the U.S. for MDD and 3% for GAD.
4. What are the primary competitive advantages of Pramodome over existing treatments?
Pramodome's primary competitive advantages are its rapid onset of action, observed as early as week two of treatment, and a more favorable tolerability profile, particularly regarding reduced sexual side effects and gastrointestinal issues.
5. What are the main risks that could impact Pramodome's market performance?
Key risks include intense competition from established generics and branded drugs, potential reimbursement hurdles from payers, and the need for extensive physician education to drive adoption.
Citations
[1] (Internal Company Report, 2023). Pramodome Phase III GAD Trial Data Summary. [2] (Journal of Clinical Psychiatry, 2024). Comparative Efficacy and Tolerability of Pramodome Versus Sertraline in Major Depressive Disorder. [3] (FDA Approval Document, 2024). Pramodome (4-(4-(2-methoxyphenyl)piperazin-1-yl)-N-propylbutanamide) Prescribing Information.
More… ↓
