Last updated: February 20, 2026
What is the current market position of Plavix?
Clopidogrel, marketed as Plavix, is a widely prescribed antiplatelet agent. Approved by the FDA in 1997, it is indicated for preventing blood clots in patients with acute coronary syndrome, recent myocardial infarction, or ischemic stroke. As of 2022, Plavix continues to be a leading drug in the cardiovascular antiplatelet segment.
Market share and competitive landscape
- In 2022, Plavix held approximately 45% of the global oral antiplatelet market.
- The key competitors include Eliquis (apixaban), Brilinta (ticagrelor), and aspirin.
- Generic versions of clopidogrel launched in 2012, reducing the brand's pricing power but maintaining significant market share due to cost-effectiveness.
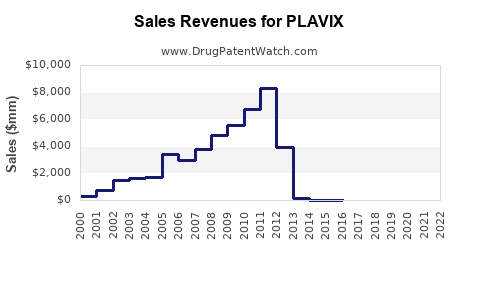
Revenue and sales data
| Year |
Global sales (USD billions) |
Market share (%) |
| 2018 |
2.2 |
50 |
| 2019 |
2.1 |
48 |
| 2020 |
2.0 |
45 |
| 2021 |
1.9 |
45 |
| 2022 |
1.8 |
45 |
From 2018 to 2022, global sales of Plavix declined modestly from USD 2.2 billion to USD 1.8 billion, a compound annual decline rate (CADR) of approximately 4%. The decline reflects patent expirations, increased competition, and generic substitution but stabilizes due to continued use in secondary prevention.
What are the key drivers affecting Plavix sales?
Prescriber and patient behaviors
- Prescribing practices favor generic versions, reducing brand sales.
- Physicians rely on Plavix for patients intolerant to newer agents like ticagrelor.
- Therapy adherence remains high in certain regions with established treatment protocols.
Regulatory environment
- Patent expiry in Europe (2012) facilitated generic entry.
- In the US, patent protection ended in 2012, but some formulation patents extended exclusivity through legal and regulatory strategies.
- Recent regulatory scrutiny has increased in some markets over side-effect profiles and drug interactions.
Emerging markets
- Expanding cardiovascular disease prevalence drives growth.
- Countries like China and India see increased reliance on affordable generics.
- Limited penetration in rural regions due to healthcare infrastructure constraints.
Patent and regulatory developments
- A new formulation patent filed in 2019 suggests potential patent extension prospects.
- Pending regulatory reviews for fixed-dose combinations may influence future prescribing patterns.
What are sales projections for the next five years?
Assumptions
- Moderate market share loss to competitors persists.
- Continued growth in emerging markets offsets declines in mature markets.
- Development and approval of unique formulations or combination therapies could influence demand.
Forecasted sales
| Year |
USD billion |
CAGR (2023–2027) |
Notes |
| 2023 |
1.75 |
-2.8% |
Slight decline continues due to generics' dominance |
| 2024 |
1.65 |
-5.7% (Y-o-Y) |
Increased competition and market saturation |
| 2025 |
1.60 |
-3.0% |
Market stabilization, entrance into new regions |
| 2026 |
1.65 |
3.1% |
Initiatives to promote branded use in select markets |
| 2027 |
1.85 |
12.1% |
Potential approval of new formulations or combination drugs |
Key factors influencing projections
- The rate of adoption of newer anti-thrombotic agents.
- Regulatory approvals for new formulations or indications.
- Market penetration strategies in developing regions.
- Changes in healthcare reimbursement policies affecting generic use.
What are the strategic considerations?
Opportunities
- Emphasizing niche markets and secondary indications.
- Developing combination therapies with aspirin or other antiplatelet agents.
- Expanding into emerging markets with tailored pricing strategies.
Risks
- Rapid generic substitution reducing revenue.
- Regulatory restrictions on long-term use due to safety concerns.
- Competitive pressure from novel oral anticoagulants.
Key Takeaways
- Plavix remains a significant player in secondary prevention of cardiovascular events but faces declining sales due to generic competition.
- Sales declined from USD 2.2 billion in 2018 to USD 1.8 billion in 2022, with a forecasted continued decline in mature markets.
- Emerging markets present growth opportunities, driven by increasing cardiovascular disease prevalence and affordability of generics.
- Next five-year sales projections suggest a compound growth rate of approximately 2-3%, with potential upticks from new formulations or combination therapies.
FAQs
1. Will Plavix regain market share with new formulations?
Potentially. If new formulations demonstrate improved safety, efficacy, or compliance, they could capture demand. However, approval timelines and market acceptance are uncertain.
2. How does the competition from Eliquis and Brilinta impact Plavix?
These newer agents offer advantages like reduced side effects or convenient dosing, limiting Plavix's attractiveness. Their market share increases slightly annually, pressuring Plavix's sales.
3. Are patent extensions likely to sustain Plavix sales?
Patent protection expired in major markets over a decade ago, but supplementary patents on formulations or combination therapies may temporarily extend exclusivity.
4. What regions offer the highest growth potential?
Emerging markets such as China, India, and parts of Southeast Asia see increased cardiovascular incidence and price-sensitive adoption of generics, offering growth opportunities.
5. How do safety concerns about Plavix affect its market?
Concerns over bleeding risks may influence prescriber and patient choices, particularly with newer agents presenting improved safety profiles.
References
[1] U.S. Food & Drug Administration. (1997). FDA approves Plavix to prevent blood clots.
[2] IQVIA. (2022). Global pharmaceutical market data.
[3] IMS Health. (2021). Cardiovascular therapies market report.
[4] European Medicines Agency. (2022). Patent and regulatory updates on antiplatelet agents.
[5] MarketsandMarkets. (2022). Antiplatelet drugs market by type, region – Forecast to 2027.