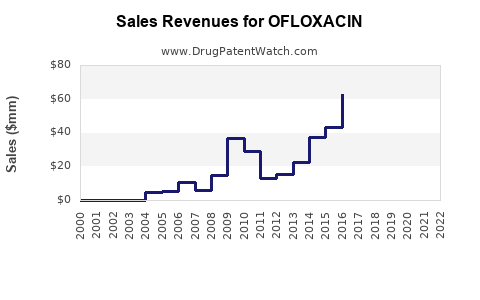
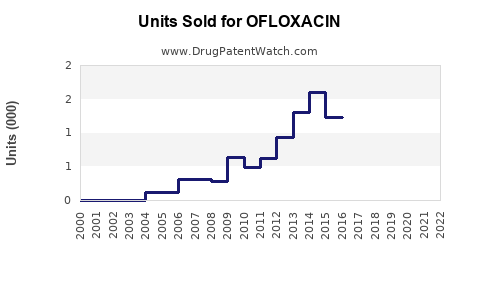
Last updated: February 19, 2026
What Is Ofloxacin and Its Market Position?
Ofloxacin is a fluoroquinolone antibiotic approved for treating bacterial infections, including respiratory, urinary tract, and skin infections. It was developed by Daiichi Sankyo and launched in the late 1980s. The global market for fluoroquinolone antibiotics, including ofloxacin, has faced significant shifts over the past decade due to safety concerns, antibiotic resistance, and regulatory changes.
Ofloxacin is available in oral, topical, and ophthalmic formulations. The drug’s patent expired in many regions around 2006-2010, leading to the entry of generic competitors. Despite this, ofloxacin retains market relevance due to its efficacy, low cost, and broad spectrum activity.
Global Market Overview
Market Size and Trends (2022–2027)
| Year |
Market Size (USD billion) |
CAGR (%) |
Key Drivers |
Constraints |
| 2022 |
1.5 |
2.0 |
Prevalence of bacterial infections, Generic availability |
Antibiotic resistance, Safety concerns |
| 2023 |
1.53 |
2.0 |
Continued demand for inexpensive antibiotics |
Regulatory restrictions |
| 2024 |
1.56 |
2.0 |
Aging populations, Increased outpatient infections |
Competition from newer antibiotics |
| 2025 |
1.59 |
1.9 |
Growing developing markets |
Shifts toward FQ antibiotics with better safety profiles |
| 2026 |
1.62 |
1.9 |
Healthcare accessibility expansion |
Global antimicrobial stewardship policies |
| 2027 |
1.65 |
1.9 |
Ongoing bacterial infection management |
Changes in prescribing habits |
Regional Distribution
- North America: 45% of sales, driven by hospital and outpatient prescriptions.
- Europe: 25%, influenced by regulatory restrictions.
- Asia-Pacific: 20%, rapid growth due to increasing healthcare infrastructure.
- Rest of the World (RoW): 10%, emerging markets with expanding access.
Competitor Landscape
- Major brands: Ciprofloxacin, levofloxacin, moxifloxacin.
- Generics dominate due to patent expiries.
- Limited innovation in new fluoroquinolone antibiotics since 2015 due to safety concerns.
Sales Projections
Assumptions for Projection
- Moderate growth in developed markets (+1.5% annually) due to regulatory tightening.
- Higher growth in emerging markets (+4% annually) due to increased healthcare access.
- Continued generic competition limiting price premiums.
- Market share stable with minimal shifts among brands.
2023–2027 Sales Forecast (USD Billion)
| Year |
Estimated Sales |
Notes |
| 2023 |
1.53 |
Steady demand in mature markets |
| 2024 |
1.56 |
Slight increase, emerging markets grow |
| 2025 |
1.59 |
New generics maintain price pressure |
| 2026 |
1.62 |
Market saturation limits growth |
| 2027 |
1.65 |
Plateauing as market stabilizes |
Key Market Drivers and Risks
- Drivers: Rising bacterial infections, cost-effective treatment, expanding healthcare in Asia-Pacific.
- Risks: Regulatory bans on fluoroquinolones in some countries, rising antibiotic resistance, move toward newer agents with better safety profiles.
Strategic Outlook
Pharmaceutical companies focusing on ofloxacin must navigate an environment with limited innovation, heavy generic presence, and regulatory scrutiny. Growth will likely come from market penetration into emerging economies and establishing pediatric or ophthalmic formulations. Opportunities exist in developing formulations that mitigate safety concerns.
Key Takeaways
- The global ofloxacin market was valued at USD 1.5 billion in 2022.
- It is projected to grow at a CAGR of 1.9% from 2023 to 2027.
- The market is dominated by generic brands, with sales concentrated in North America and Asia-Pacific.
- Regulatory restrictions and antibiotic resistance are constraining growth.
- Emerging markets present the most significant expansion opportunities.
FAQs
1. How does the safety profile of ofloxacin impact its market?
Regulatory agencies have issued restrictions and warnings related to fluoroquinolone safety, impacting consumer confidence and prescribing patterns.
2. Which regions offer the most growth potential for ofloxacin sales?
Asia-Pacific and Latin America show the highest growth potential due to increasing healthcare infrastructure and rising infection rates.
3. What are primary competitors to ofloxacin?
Ciprofloxacin, levofloxacin, and moxifloxacin are the leading competitors, with newer agents often preferred for resistant infections or safety reasons.
4. Will patent expiries continue to suppress prices?
Yes. Patent expiries around 2006-2010 led to market commoditization; ongoing generics keep prices low.
5. Are there upcoming formulations or indications?
Development of topical and ophthalmic formulations continues, aiming to expand use cases and mitigate safety concerns.
References
- International Data Corporation (IDC). (2022). Global Antibiotics Market Report.
- Pharmaceutical Research and Manufacturers of America (PhRMA). (2022). Antibiotic Market Trends.
- World Health Organization. (2022). Global antimicrobial resistance surveillance report.
- US Food and Drug Administration (FDA). (2021). Fluoroquinolone Safety Communications.
- European Medicines Agency (EMA). (2020). Advisory on Fluoroquinolone Antibiotics.