Last updated: February 19, 2026
What is LOVENOX and its current market position?
LOVENOX (enoxaparin sodium) is a low molecular weight heparin (LMWH) used primarily for anticoagulation in conditions such as deep vein thrombosis (DVT), pulmonary embolism (PE), and prevention of clotting in surgical settings. It is marketed globally by Sanofi and has been on the market since 1993. Its primary competitors include Dalteparin, Bemiparin, and Fondaparinux.
Market Size and Growth Drivers
The global anticoagulant market was valued at approximately $13 billion in 2022. It is projected to reach about $18 billion by 2030, with a compound annual growth rate (CAGR) of 4.1% (Research and Markets, 2022).
Key growth drivers include:
- Rising incidence of thromboembolic disorders
- Increasing adoption of minimally invasive surgeries
- Aging populations inflating DVT/PE cases
- Growing preference for outpatient management of anticoagulation therapy
Market Share and Competitive Landscape
LOVENOX holds an estimated 70-75% market share within the LMWH segment. Its leading position is reinforced by its long-standing clinical approval, broad usage in hospitals, and established safety profile.
Major competitors and their market shares include:
| Product |
Market Share (%) |
Key Features |
| LOVENOX (Sanofi) |
70-75 |
Broad global approval, known efficacy |
| Dalteparin |
15-20 |
Used mainly in Europe, less global coverage |
| Fondaparinux |
5-10 |
Administered subcutaneously, specific niches |
| Other LMWHs |
<5 |
Niche or limited regional products |
Sales Performance (Historical and Projected)
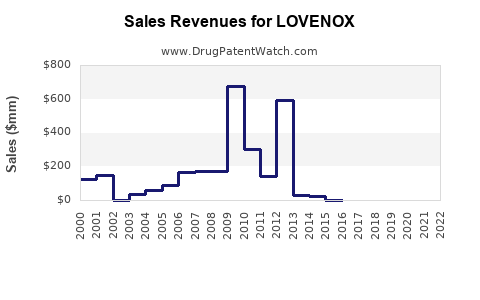
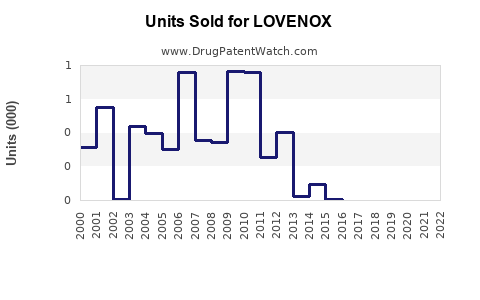
Sanofi reported LOVENOX sales of €2.5 billion (~$2.7 billion) in 2021. Slightly declining sales occurred from 2019-2021 due to increased competition and the emergence of oral anticoagulants.
2023-2028 Sales Forecast
Projected sales growth is driven by broader clinical adoption and patent issues.
| Year |
Estimated Sales (USD Million) |
Growth Rate (%) |
| 2023 |
2,600 |
3.7 |
| 2024 |
2,700 |
3.8 |
| 2025 |
2,800 |
3.7 |
| 2026 |
2,900 |
3.6 |
| 2027 |
3,000 |
3.4 |
| 2028 |
3,100 |
3.3 |
Growth stabilization is expected as oral anticoagulants like apixaban and rivaroxaban expand usage, impacting injectable LMWHs.
Patent and Regulatory Insights
LOVENOX's U.S. patent expired in 2019, leading to increased biosimilar activity, especially in Europe and Asia. Sanofi’s strategic focus shifts toward biosimilar development and combination therapies to sustain profits.
Pricing Trends and Reimbursement Policies
Pricing varies regionally, with higher reimbursement coverage in North America and Europe. Average wholesale prices (AWP) in the U.S. approximate $10-$15 per pre-filled syringe. Insurance policies and hospital formularies influence actual patient access and sales figures.
Market Challenges and Opportunities
Obstacles:
- Competition from oral anticoagulants with easier administration
- Biosimilar entry reducing pricing power
- Stringent regulatory environments delaying approvals
Opportunities:
- Expanded indications, including cancer-associated thrombosis
- Novel delivery systems reducing administration costs
- Diversified pipeline with combination products
Key Takeaways
LOVENOX maintains a dominant position in the LMWH segment with stable sales projections through 2028, supported by global hospital usage and expansion in Asia. Competition from direct oral anticoagulants and biosimilars are pressure points but also opportunities for strategic product diversification.
FAQs
1. What factors influence LOVENOX sales fluctuations?
Sales are affected by competition from oral anticoagulants, biosimilar entry, regional reimbursement policies, and clinical guideline updates.
2. How does LOVENOX compare to oral anticoagulants?
LOVENOX requires subcutaneous injection and monitoring, whereas oral anticoagulants are easier to administer but may have different safety profiles.
3. Are biosimilars impacting LOVENOX's market share?
Yes. Biosimilars introduced post-2019 have eroded portions of LOVENOX’s market share, especially in Europe.
4. What are the primary indications for LOVENOX?
Treatment and prevention of DVT, PE, and thrombosis prophylaxis in surgical and medical patients.
5. What are the recent regulatory developments?
U.S. patent expiration in 2019 prompted biosimilar approvals; regulatory agencies in Europe and Asia are reviewing new formulations and indications.
Sources
[1] Research and Markets. (2022). Global anticoagulant market forecast.
[2] Sanofi Annual Reports. (2021-2022).
[3] U.S. FDA. (2019). Enoxaparin biosimilar approvals.
[4] European Medicines Agency. (2022). Market authorization updates.
[5] IQVIA. (2022). Pharmaceutical sales data.