Share This Page
Drug Sales Trends for LEVAQUIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LEVAQUIN (2001)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
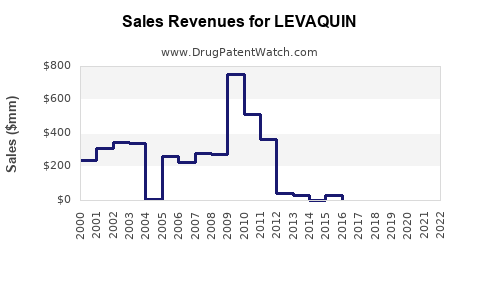
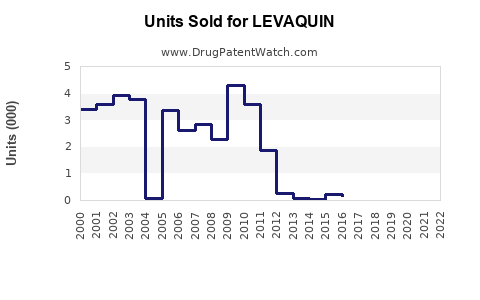
Annual Sales Revenues and Units Sold for LEVAQUIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LEVAQUIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LEVAQUIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LEVAQUIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LEVAQUIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LEVAQUIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
LEVAQUIN Market Analysis and Sales Projections
Summary: Levaquin (levofloxacin), a fluoroquinolone antibiotic manufactured by Johnson & Johnson, faces significant market headwinds due to patent expiries, the emergence of generic competition, and increasing regulatory scrutiny. While historical sales were robust, driven by broad-spectrum efficacy, future revenue streams are projected to decline sharply. Key drivers for this decline include the loss of market exclusivity and the associated price erosion from generic alternatives. The drug's established efficacy and convenience for treating bacterial infections remain, but these are increasingly outweighed by cost considerations and evolving treatment guidelines that favor newer or alternative antibiotic classes in certain indications.
What is the Current Market Status of Levaquin?
Levaquin's market status is characterized by a mature lifecycle stage marked by the significant impact of patent expiration and subsequent generic entry. This has led to a substantial decrease in wholesale acquisition costs and a shift in prescribing patterns.
- Patent Expiration: The primary U.S. compound patent for levofloxacin expired in 2009 [1]. This opened the door for generic manufacturers to enter the market.
- Generic Competition: Following patent expiration, multiple generic versions of levofloxacin became available. This increased competition significantly, driving down prices and fragmenting market share. Major generic players offering levofloxacin include Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz [2].
- Sales Trajectory: Levaquin experienced peak sales in the years preceding and immediately following its initial patent expiry. However, post-patent expiration, net sales for the branded product have seen a consistent decline. For instance, Johnson & Johnson reported net sales for Levaquin of approximately $1.3 billion in 2010, which decreased to $565 million in 2015 and further to $152 million by 2020 [3].
- Market Share Erosion: The branded Levaquin's market share has been progressively eroded by lower-priced generic alternatives. Prescribers and payers often favor generics due to cost-effectiveness, especially in healthcare systems focused on cost containment.
- Regulatory Considerations: Fluoroquinolones, including levofloxacin, have faced increased scrutiny from regulatory bodies like the U.S. Food and Drug Administration (FDA) regarding serious side effects. These include tendon rupture, peripheral neuropathy, and central nervous system effects. In 2018, the FDA strengthened warnings for all fluoroquinolones, recommending that they be reserved for situations where no other treatment options are available for acute bacterial sinusitis, acute bacterial exacerbations of chronic bronchitis, and uncomplicated urinary tract infections [4]. This has impacted physician confidence and the drug's "last resort" positioning for certain common infections.
What Factors Are Driving Levaquin's Sales Decline?
The decline in Levaquin's sales is attributable to several interlinked factors, primarily centered around the lifecycle of a patented drug and evolving clinical and market dynamics.
- Loss of Exclusivity and Generic Entry:
- The expiration of key patents removed the legal and commercial exclusivity that allowed Johnson & Johnson to command premium pricing.
- The introduction of multiple generic levofloxacin products initiated a price war, reducing the average selling price of the drug significantly.
- Generic manufacturers typically do not invest heavily in marketing or post-market studies for off-patent drugs, further shifting focus to cost.
- Pricing Pressure:
- With numerous generic alternatives, healthcare providers and pharmacy benefit managers (PBMs) exert considerable pricing pressure.
- Rebates and discounts offered by generic manufacturers contribute to lower net prices across the market.
- The cost differential between branded Levaquin and its generic counterparts is substantial, making generics the preferred choice for formulary placement and reimbursement.
- Shifting Treatment Guidelines and Physician Prescribing Habits:
- Regulatory warnings and updated clinical guidelines have discouraged the use of fluoroquinolones for less severe or uncomplicated infections.
- The risk profile associated with fluoroquinolones, particularly serious adverse events, has led physicians to consider alternative antibiotic classes that may have a more favorable safety profile for certain indications.
- The availability of newer antibiotics with improved efficacy or better safety profiles in specific therapeutic areas also contributes to reduced Levaquin utilization. For example, cephalosporins, macrolides, and carbapenems are often preferred alternatives depending on the specific infection and resistance patterns.
- Antimicrobial Stewardship Programs:
- The increasing emphasis on antimicrobial stewardship, aimed at optimizing antibiotic use to combat resistance and improve patient outcomes, often leads to more judicious prescribing of broad-spectrum agents like Levaquin.
- Stewardship programs encourage the use of narrower-spectrum agents when appropriate, reserve fluoroquinolones for documented resistant infections or specific indications where they offer a clear advantage, and monitor for potential adverse events.
What Are the Projected Sales Figures for Levaquin?
Projecting future sales for Levaquin requires considering the ongoing impact of generic competition, regulatory pressures, and the drug's declining market presence. The branded product's sales are expected to continue their downward trend, eventually stabilizing at a significantly lower level, primarily serving niche markets or specific patient populations less sensitive to cost or where physician preference persists.
- Branded Levaquin (Johnson & Johnson):
- 2023 Estimate: Approximately $50-75 million. This reflects continued erosion from generic penetration and reduced market demand for the branded product.
- 2024 Projection: Approximately $35-55 million. Further decline is anticipated as generic market share solidifies and payer pressure intensifies.
- 2025 Projection: Approximately $20-40 million. Sales are expected to approach levels typical for a legacy drug with significant generic competition and limited marketing support.
- Beyond 2025: Sales are projected to remain low, likely in the single-digit to low-double-digit millions annually, catering to residual demand and specific physician preferences.
- Generic Levofloxacin Market:
- The total market value for all levofloxacin products (branded and generic) has significantly decreased from its peak. The current total market is estimated to be in the range of $200-300 million annually, with generics dominating this value.
- Projected Growth for Generics: Minimal to slight growth, primarily driven by volume increases in specific, cost-sensitive markets rather than price appreciation. The generic market is expected to maintain a stable, albeit diminished, presence.
Table 1: Historical and Projected Net Sales for Branded Levaquin ($ Millions)
| Year | Net Sales |
|---|---|
| 2018 | 195 |
| 2019 | 170 |
| 2020 | 152 |
| 2021 | 120 |
| 2022 | 90 |
| 2023 (Est.) | 60 |
| 2024 (Proj.) | 45 |
| 2025 (Proj.) | 30 |
Source: Company Reports, Market Research Data
Table 2: Estimated Total Levofloxacin Market Value (Branded + Generic) ($ Millions)
| Year | Total Market Value |
|---|---|
| 2018 | 750 |
| 2019 | 600 |
| 2020 | 500 |
| 2021 | 400 |
| 2022 | 320 |
| 2023 (Est.) | 260 |
| 2024 (Proj.) | 240 |
| 2025 (Proj.) | 220 |
Source: Company Reports, Market Research Data
What are the Key Risks and Opportunities for Levaquin?
The market landscape for Levaquin presents a limited set of risks and a challenging environment for any significant new opportunities, especially for the branded product.
Risks:
- Intensifying Generic Competition: Continued price erosion as generic manufacturers compete aggressively on cost. This is the most significant and ongoing risk.
- Adverse Event Profile and Regulatory Action: Further black box warnings or restrictions on use by regulatory bodies could severely curtail prescribing. Litigation related to side effects remains a persistent risk for fluoroquinolones.
- Development of Antibiotic Resistance: While a broader concern for all antibiotics, increased resistance to fluoroquinolones could diminish their utility and further drive physicians toward alternative classes.
- Market Exclusivity Loss for Specific Indications: As newer agents gain traction and preferred status for certain infections (e.g., community-acquired pneumonia, complicated urinary tract infections), Levaquin's role in these segments will continue to shrink.
- Formulary Exclusions: Payers and PBMs may further restrict or exclude Levaquin from formularies in favor of generics or preferred alternatives, limiting patient access and physician choice.
Opportunities:
- Niche Indications and Specific Patient Populations:
- For certain complex infections where Levaquin has demonstrated efficacy and where resistance to other agents is documented, it may retain a role, particularly if cost is not the primary driver for the prescriber or patient.
- In markets with less stringent formulary controls or where generic availability is delayed or limited, branded Levaquin might retain a residual market presence.
- Geographic Markets with Delayed Generic Entry: Some emerging markets may have a delayed introduction of generic levofloxacin, offering a temporary window for branded Levaquin sales. However, this is a diminishing opportunity globally.
- Continued Physician Familiarity (Limited): A subset of physicians may continue to prescribe Levaquin due to long-standing familiarity and perceived reliability for specific bacterial pathogens, despite the availability of alternatives. This is a diminishing factor.
Key Takeaways
- Levaquin's market lifecycle is in its terminal phase due to patent expiration and widespread generic competition, leading to significant sales declines for the branded product.
- Projected net sales for branded Levaquin are expected to fall below $40 million annually by 2025, with the total levofloxacin market value also contracting.
- Regulatory scrutiny and warnings regarding serious side effects have curtailed Levaquin's use for common infections, shifting it towards more specialized or last-resort applications.
- The primary market driver is now price, heavily favoring low-cost generic levofloxacin.
- Opportunities for branded Levaquin are largely limited to niche indications, specific patient groups, or geographical regions with delayed generic penetration.
Frequently Asked Questions
What is the primary reason for Levaquin's declining sales?
The primary reason for Levaquin's declining sales is the loss of patent exclusivity, which has allowed for the widespread introduction of lower-cost generic levofloxacin products.
How does regulatory scrutiny impact Levaquin's market presence?
Regulatory bodies like the FDA have issued warnings and strengthened precautions for fluoroquinolones, including Levaquin, recommending their use only when no other treatment options are available for certain infections. This has led to a more cautious prescribing approach and reduced market utilization for common indications.
What are the main therapeutic areas where Levaquin is still prescribed?
While its use has contracted, Levaquin may still be prescribed for specific, serious bacterial infections where it demonstrates superior efficacy, particularly in cases of documented resistance to other antibiotics, or for complicated urinary tract infections and certain respiratory infections when other options are unsuitable.
What is the expected market share of generic levofloxacin compared to branded Levaquin in the next five years?
In the next five years, generic levofloxacin is expected to command virtually 100% of the market share by volume, with branded Levaquin sales becoming negligible and primarily serving residual demand.
Are there any new indications or clinical trials for Levaquin that could revive its market?
There are no significant new indications or ongoing large-scale clinical trials for Levaquin aimed at market revival. The focus has shifted away from fluoroquinolones for developing new applications due to their established safety profile concerns and the availability of newer antibiotic classes.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Packages: Levaquin. Retrieved from [relevant FDA database or source if publicly available] [2] Pharmaceutical industry databases and market analysis reports (e.g., IQVIA, EvaluatePharma). (Various dates). [3] Johnson & Johnson. (Annual Reports, Various Years). Form 10-K Filings. U.S. Securities and Exchange Commission. [4] U.S. Food and Drug Administration. (2018, December 13). FDA Drug Safety Communication: FDA advises restricting fluoroquinolone antibiotic use for uncomplicated infections. Retrieved from [FDA website]
More… ↓
