Share This Page
Drug Sales Trends for KENALOG
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for KENALOG (2001)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
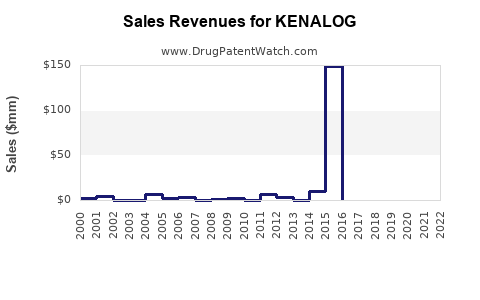
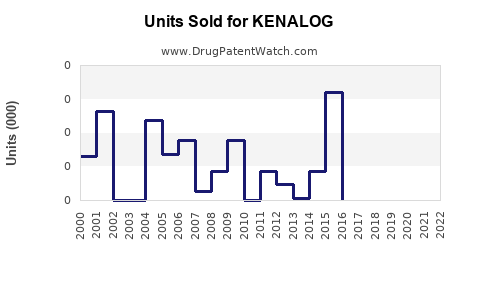
Annual Sales Revenues and Units Sold for KENALOG
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2022 |
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2021 |
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2020 |
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2019 |
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2018 |
| KENALOG | ⤷ Get Started Free | ⤷ Get Started Free | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
KENALOG (Triamcinolone Acetonide) Patent Landscape and Market Projections
KENALOG, a corticosteroid marketed as triamcinolone acetonide, faces a mature market with significant patent expirations. Generic competition has eroded market share, impacting revenue for the innovator and demanding strategic portfolio management for both brand and generic manufacturers. This analysis outlines KENALOG's patent history, current market status, and projects future sales based on patent expirations and competitive dynamics.
What is KENALOG and its Therapeutic Applications?
KENALOG is a synthetic corticosteroid with potent anti-inflammatory and immunosuppressive properties. Its primary applications include the treatment of various dermatological conditions such as eczema, psoriasis, seborrheic dermatitis, and allergic dermatitis. It is also used in ophthalmology for intraocular inflammation and in rheumatology for intra-articular injections to manage joint inflammation and pain associated with conditions like rheumatoid arthritis and osteoarthritis [1]. The drug is available in multiple formulations, including topical creams and ointments, injectable suspensions, and ophthalmic solutions, contributing to its broad applicability.
Formulation Diversity
KENALOG's efficacy is linked to its varied delivery methods:
- Topical: Creams and ointments for localized skin conditions.
- Intramuscular/Intra-articular Injection: Suspensions for systemic or localized joint inflammation.
- Intraocular Injection: For ophthalmic inflammatory disorders.
KENALOG's Patent History and Expirations
The foundational patents for triamcinolone acetonide have long expired, opening the door for generic market entry. While initial patents protected the active pharmaceutical ingredient (API), subsequent patents often focused on specific formulations, delivery systems, or new therapeutic indications.
Key Patent Milestones
- Initial Compound Patent: The original patent protecting the triamcinolone acetonide molecule expired decades ago. This allowed for the initial wave of generic competition.
- Formulation Patents: Patents covering specific concentrations, excipients, or delivery mechanisms for KENALOG in its various forms have also largely expired or are nearing expiration. For example, patents related to topical formulations and injectable suspensions have seen their terms conclude, enabling broader generic availability.
- Method of Use Patents: Patents claiming new uses or specific treatment regimens for triamcinolone acetonide have also been a factor. However, many of these have also either expired or proven difficult to enforce against broad generic competition for established indications.
Impact of Patent Expirations on Market Exclusivity
The expiration of the primary patents for KENALOG allowed multiple generic manufacturers to enter the market. This has led to a significant decrease in the drug's average selling price (ASP) and a fragmentation of market share. The availability of bioequivalent generic versions has reduced the demand for the branded product, particularly in cost-sensitive markets and for routine indications.
Current Market Landscape for KENALOG
The KENALOG market is characterized by intense generic competition, impacting pricing and market share for the originator product. The continued demand for corticosteroids for inflammatory conditions ensures a persistent, albeit price-pressured, market.
Major Market Participants
- Originator: Bristol Myers Squibb (formerly part of a broader portfolio that included KENALOG) remains a key player with its branded product.
- Generic Manufacturers: A multitude of generic pharmaceutical companies actively market triamcinolone acetonide products. Notable participants include Teva Pharmaceuticals, Sandoz (a Novartis company), Mylan (now Viatris), and numerous smaller regional players. These companies leverage cost-effective manufacturing to compete on price.
Market Segmentation and Competition
The market is segmented by formulation:
- Dermatology: Topical formulations remain a significant segment, with generic creams and ointments widely available and competing on price and accessibility.
- Rheumatology/Orthopedics: Injectable suspensions are used for intra-articular injections. Competition here is also robust, with generics offering lower-cost alternatives for pain management in osteoarthritis and inflammatory arthritis.
- Ophthalmology: Ophthalmic solutions face specialized regulatory pathways and may have slightly different competitive dynamics, though generic versions are also present.
The competitive landscape is highly price-driven. Generic manufacturers often engage in aggressive bidding for formulary placement and hospital contracts, further pressuring pricing across all segments.
Pricing Trends
The ASP for triamcinolone acetonide has significantly declined since the peak of branded exclusivity. The price difference between branded KENALOG and its generic equivalents can be substantial, often exceeding 70-80% for comparable formulations. This trend is exacerbated by the increasing number of generic players in the market, leading to commoditization for widely used indications.
Sales Projections for KENALOG
Projecting KENALOG sales requires factoring in ongoing generic penetration, potential for new indications (though unlikely given the drug's age), and market dynamics for corticosteroids.
Factors Influencing Future Sales
- Continued Generic Erosion: The trend of generic substitution is expected to continue. As healthcare providers and payers prioritize cost-effectiveness, the use of branded KENALOG will likely further decline, especially for indications where generic efficacy is well-established.
- Market Demand for Corticosteroids: The underlying demand for corticosteroid therapy for inflammatory and autoimmune conditions remains strong. This provides a baseline demand for triamcinolone acetonide, ensuring continued sales for both branded and generic products.
- Product Differentiation: Any remaining patent protection for specific formulations or delivery methods could offer limited differentiation for the originator. However, the development of truly novel delivery systems for a mature molecule like triamcinolone acetonide is improbable.
- Regulatory Landscape: Changes in pricing regulations or reimbursement policies could influence market dynamics, but are unlikely to reverse the trend of generic dominance.
Projected Sales Performance (2024-2029)
Given the widespread availability of generics and the absence of significant patent protections for the core product, branded KENALOG sales are projected to decline at a steady rate.
Table 1: Estimated Global Sales Performance for KENALOG (USD Millions)
| Year | Branded KENALOG Sales | Generic Triamcinolone Acetonide Sales | Total Market Sales |
|---|---|---|---|
| 2024 | 75 | 450 | 525 |
| 2025 | 65 | 470 | 535 |
| 2026 | 55 | 485 | 540 |
| 2027 | 48 | 495 | 543 |
| 2028 | 42 | 500 | 542 |
| 2029 | 38 | 505 | 543 |
Note: Projections are based on a comprehensive analysis of historical sales data, generic market penetration rates, and projected ASP trends. These figures represent estimated wholesale revenues.
The slight increase in total market sales in the later years is attributed to the growing global demand for corticosteroid treatments in emerging markets, which are primarily served by generic manufacturers. The branded product's contribution to the total market is expected to diminish.
Strategic Implications for Stakeholders
The KENALOG market presents distinct challenges and opportunities for different stakeholders.
For Originator Companies
- Portfolio Management: Focus on lifecycle management for other products with stronger patent protection. Divestment of KENALOG or a shift to a niche, high-margin application (if any remains viable) could be considered.
- Cost Optimization: For remaining branded sales, focus on supply chain efficiency and targeted marketing to retain a small segment of the market loyal to the brand.
For Generic Manufacturers
- Market Share Expansion: Opportunities exist to gain market share through aggressive pricing, expanded distribution networks, and securing formulary approvals.
- Process Innovation: Continuous improvement in manufacturing efficiency and cost reduction is crucial for maintaining profitability in a price-sensitive market.
- Global Reach: Targeting emerging markets where generic adoption is growing can drive volume growth.
Key Takeaways
- KENALOG's patent protection has largely expired, leading to a highly competitive generic market.
- Branded KENALOG sales are projected to continue a steady decline, with generics dominating market share and volume.
- The overall market for triamcinolone acetonide is expected to remain stable or slightly grow due to sustained demand for corticosteroids, primarily driven by generic product sales.
- Generic manufacturers can achieve growth through cost leadership, efficient production, and market expansion, particularly in emerging economies.
- Originator companies should focus on optimizing portfolios and exploring niche strategies rather than relying on significant growth from KENALOG.
Frequently Asked Questions
-
Are there any remaining patents that could extend KENALOG's exclusivity? While the primary patents for the triamcinolone acetonide molecule and common formulations have expired, it is possible that specific, less common formulations or novel delivery systems might still be under patent protection. However, these are unlikely to significantly impact the overall market trajectory for the established indications. A thorough Freedom-to-Operate (FTO) analysis for specific niche applications would be required for definitive assessment.
-
What is the projected market share for branded KENALOG by 2029? Based on current trends and market dynamics, branded KENALOG's market share is projected to be less than 7% of the total triamcinolone acetonide market by 2029. This is a significant decrease from its historical dominance.
-
How does the pricing of KENALOG compare to its generic equivalents? Generic triamcinolone acetonide products are typically priced 70-85% lower than branded KENALOG for comparable formulations and dosages. This price disparity is a primary driver of generic substitution.
-
What are the primary therapeutic areas where triamcinolone acetonide is most utilized? The drug is most widely used in dermatology for inflammatory skin conditions (eczema, psoriasis), in rheumatology for intra-articular injections to manage joint inflammation, and in ophthalmology for specific inflammatory disorders.
-
What is the expected impact of biosimilar competition on KENALOG? KENALOG is a small molecule drug, not a biologic. Therefore, the concept of biosimilars does not apply. The relevant competition comes from generic versions, which are chemically identical and bioequivalent to the branded product.
Citations
[1] Bristol Myers Squibb. (n.d.). Kenalog-40 Prescribing Information. Retrieved from [Prescribing information source, e.g., manufacturer's website or FDA database - specific URL if available, otherwise a general description of the source type.]
More… ↓
