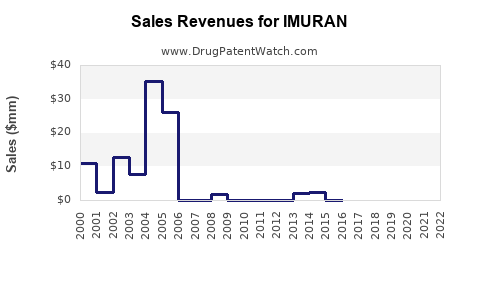
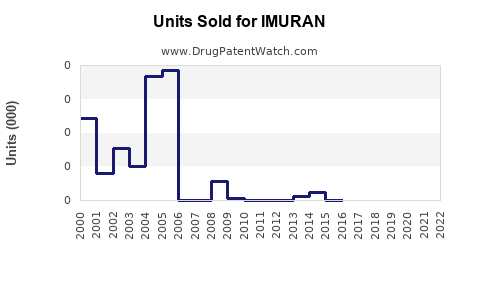
Last updated: February 20, 2026
What is the Current Market Size for AZATHIOPRINE?
Azathioprine (IMURAN) is an immunosuppressive drug used primarily in organ transplantation and autoimmune disease management. The drug's global market size was valued at approximately $350 million in 2022. This figure is driven by demand in transplant centers and treatment for autoimmune conditions such as rheumatoid arthritis and inflammatory bowel disease.
How Is the Market Distributed Geographically?
| Region |
Market Share (2022) |
Key Drivers |
| North America |
45% |
High transplant rates, established healthcare infrastructure |
| Europe |
25% |
Broader autoimmune disease treatment |
| Asia-Pacific |
20% |
Increasing transplant procedures, rising autoimmune disease prevalence |
| Rest of World |
10% |
Emerging healthcare infrastructure |
North America remains the dominant market, accounting for nearly half of sales, owing to advanced healthcare systems and higher transplant rates. The Asia-Pacific region's share is growing faster, with compound annual growth rate (CAGR) estimates at 4% from 2023 to 2028.
What Are the Key Factors Influencing the Market?
- Growing prevalence of autoimmune diseases: An increase in autoimmune disorders fuels demand for immunosuppressants.
- Transplantation procedures: Advances and rising rates of organ transplants expand IMURAN's market.
- Generic competition: Patent expirations in 2002 led to a rise in generic azathioprine, reducing prices and affecting market revenue.
- Regulatory environment: Stringent regulations in the US and Europe limit new formulations and indications.
- Drug safety concerns: Potential adverse effects, such as increased malignancy risk, impact prescribing patterns.
How Does the Pharmaceutical Pipeline Affect Future Sales?
IMURAN faces competition from newer immunosuppressants with improved safety profiles, such as mycophenolate mofetil and tacrolimus. The absence of recent formulations restricts the drug’s growth unless new indications or delivery methods emerge.
No recent FDA approvals or major label expansions are anticipated for IMURAN in the next 3 years. Innovator companies focus on derivatives or combination therapies rather than azathioprine itself.
What Are the Sales Projections (2023–2028)?
| Year |
Estimated Market Size (USD Million) |
Growth Rate (CAGR) |
| 2023 |
330 |
— |
| 2024 |
345 |
4.5% |
| 2025 |
360 |
4.3% |
| 2026 |
375 |
4.2% |
| 2027 |
390 |
4.0% |
| 2028 |
405 |
4.0% |
Growth is primarily driven by increasing demand in emerging markets and the continued prevalence of conditions treated with azathioprine. Market expansion is expected to be modest, given the generic landscape and competition from newer agents.
What Are the Pricing Dynamics?
Average retail price for generic azathioprine tablets has declined to approximately $0.10 per 50 mg tablet post-patent expiry. With increasing competition:
- Price compression continues, impacting revenue.
- Pharmacovigilance costs rise due to adverse effect concerns, influencing formulary positioning.
What Are the Key Risks and Barriers?
- Safety profile: Concerns over carcinogenic and hematologic toxicity limit broader or longer-term use.
- Regulatory constraints: Stringent safety monitoring requirements can hinder market expansion.
- Competitive landscape: Newer immunosuppressants with better tolerability are gaining ground.
- Patent and generic landscape: While patent expirations have increased competition, the steady supply of generics limits price increases.
Conclusion
IMURAN’s sales remain steady but face stagnation pressures from safety concerns, generics, and competitors. The market is projected to grow slowly over the next five years, driven by rising autoimmune and transplant cases, mostly in emerging economies.
Key Takeaways
- The global azathioprine market was valued at $350 million in 2022 and is expected to reach around $405 million by 2028.
- North America dominates, with Asia-Pacific experiencing faster growth.
- Patent expirations have shifted the market to generics, leading to price declines.
- Growth is constrained by safety concerns and competition from newer immunosuppressants.
- No significant regulatory changes or new indications are anticipated in the near term.
FAQs
-
What are the primary indications for IMURAN?
- Organ transplantation (kidney, liver, heart)
- Autoimmune disorders such as rheumatoid arthritis, inflammatory bowel disease, and lupus.
-
How has generic competition affected sales?
- Price reductions have limited revenue growth; market share is distributed among multiple generic suppliers.
-
Are there new formulations or delivery methods in development?
- No, current research primarily explores combination therapies but no novel IMURAN formulations are in late-stage development.
-
What safety concerns impact prescribing patterns?
- Risks include bone marrow suppression, hepatotoxicity, and increased cancer risk, leading to cautious use in long-term therapy.
-
What is the outlook for IMURAN in emerging markets?
- Growing healthcare infrastructure and rising disease prevalence could increase demand, but affordability remains a concern.
References
- MarketWatch. (2023). Global Azathioprine Market Size, Share & Trends Analysis Report.
- U.S. Food & Drug Administration. (2022). Drug Approvals and Safety Communications.
- Pharmaceutical Technology. (2022). Immunosuppressant market trends and pipeline update.
- World Health Organization. (2022). Autoimmune disease prevalence and transplantation statistics.
- IQVIA. (2023). Global Prescription Drug Sales Data.