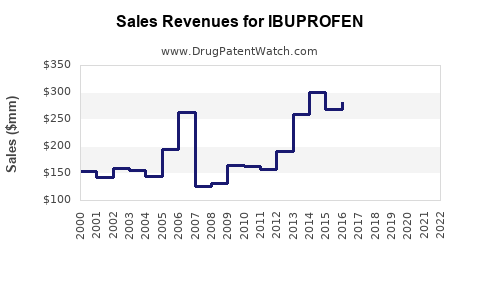
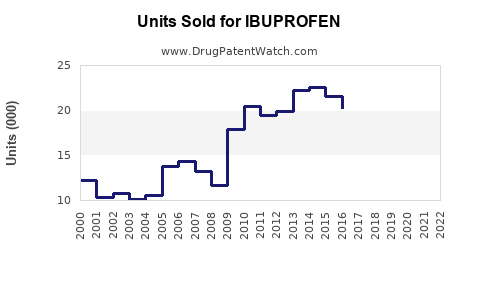
Last updated: February 12, 2026
What Is the Global Market Size for Ibuprofen?
The global ibuprofen market was valued at approximately $4.3 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2023 to 2030, reaching around $5.4 billion by 2030.
What Are the Key Drivers of Market Growth?
- Increasing prevalence of chronic pain conditions and acute pain management needs.
- Growing awareness of over-the-counter (OTC) medications for self-care.
- Rising demand in emerging markets due to expanding healthcare infrastructure.
- Regulatory approvals for new formulations, including topical and sustained-release options.
How Is the Market Segmented?
By Formulation:
- Oral tablets and capsules account for approximately 75% of sales, primarily OTC.
- Topical gels and creams account for about 20%, aiming at localized pain.
- Other forms (suspensions, powders) make up roughly 5%.
By Application:
- Pain relief (including dental, muscular, migraines): 60%.
- Fever reduction: 25%.
- Anti-inflammatory uses: 15%.
By Distribution Channel:
- OTC pharmacies: 50%.
- Hospital pharmacies: 30%.
- Online sales: 15%.
- Other channels (drug stores, departmental stores): 5%.
Who Are the Major Players?
Leading manufacturers include:
- BASF (Germany): Supplies raw materials and finished formulations.
- Johnson & Johnson (U.S.): Sells OTC products under brands like Motrin.
- Reckitt Benckiser (U.K.): Markets Nurofen in various markets.
- Amneal Pharmaceuticals (U.S.): Produces generic ibuprofen.
- Meda Pharmaceuticals (Sweden): Known for OTC pain products.
The market features both global multinationals and regional generic manufacturers, with significant growth seen in key emerging markets such as China, India, and Brazil.
What Are the Sales Projections for 2023-2030?
| Year |
Estimated Market Size (USD Billion) |
CAGR |
Notes |
| 2023 |
4.45 |
3.2% |
Estimated based on prior trends. |
| 2024 |
4.59 |
— |
Slight increase from 2023. |
| 2025 |
4.74 |
— |
Continued growth with emerging markets. |
| 2027 |
5.1 |
— |
Market expansion driven by new formulations. |
| 2030 |
5.4 |
— |
Reached projected value. |
Note: These figures are estimates based on industry reports from MarketsandMarkets, Grand View Research, and IBISWorld.
How Is the Regulatory Landscape Affecting Sales?
- FDA and EMA approvals favor OTC availability, boosting sales.
- Restrictions on certain formulations (e.g., high-dose ibuprofen in some regions) influence sales volume.
- Patent expirations for branded formulations have opened opportunities for generic manufacturers, increasing market competition and lowering prices.
What Are the Key Opportunities and Challenges?
Opportunities:
- Expansion into emerging markets with low OTC penetration.
- Development of long-acting or targeted delivery forms.
- Integration of digital health tools for OTC sales.
Challenges:
- Regulatory hurdles delaying new formulations.
- Consumer safety concerns leading to tighter regulations.
- Competition from other NSAIDs like naproxen and aspirin.
Key Takeaways
- The ibuprofen market is growing steadily, driven by the rising need for pain and fever management.
- Oral formulations dominate, but topical and other forms present new growth avenues.
- Major companies are expanding product lines and market reach, especially in emerging economies.
- Regulatory policies significantly influence market dynamics, favoring OTC sales but imposing restrictions.
- Generics are contributing to lower prices but increasing competition.
FAQs
1. How does ibuprofen compare with other NSAIDs in sales?
Ibuprofen holds the largest share among OTC NSAIDs, with global sales higher than naproxen and aspirin, primarily because of its extensive OTC availability.
2. What are the major concerns impacting ibuprofen sales?
Safety concerns such as gastrointestinal risks and cardiovascular side effects influence regulatory oversight and consumer acceptance.
3. What innovations are expected in the ibuprofen market?
Long-acting formulations, topical gels, and combination products with other analgesics are under development.
4. How does the rise of online pharmacies affect sales?
Online channels facilitate easier access, especially in markets with limited OTC infrastructure, contributing to sales growth.
5. Which geographic region presents the most growth opportunity?
Emerging markets like India and China, where OTC analgesics are gaining popularity, offer significant growth potential.
References
- MarketsandMarkets. "NSAID Market by Product, Application, Distribution Channel," 2022.
- Grand View Research. "Analgesics Market Size, Share & Trends," 2023.
- IBISWorld. "Pain Management Pharmaceutical Industry Outlook," 2023.