Last updated: February 14, 2026
What is GEODON (Ziprasidone) and its approved indications?
GEODON (ziprasidone) is an atypical antipsychotic primarily indicated for the treatment of schizophrenia and acute agitation associated with schizophrenia. It is also approved for bipolar disorder, specifically for episodes of mania or mixed episodes. The drug was first approved by the U.S. Food and Drug Administration (FDA) in 2001.
How is the market for GEODON structured?
The market comprises several segments:
- Schizophrenia treatment: The largest segment, driven by the prevalence of schizophrenia, estimated at 1% globally, with 2.4 million Americans diagnosed.
- Bipolar disorder: Comprises about 2.8% of adults, with recent growth in long-term management.
- Acute agitation management: A smaller but significant section, mainly in inpatient settings.
The drug faces competition from other atypical antipsychotics such as risperidone, olanzapine, quetiapine, and aripiprazole. Generic versions of some competitors affect GEODON’s market share.
What are the current sales figures for GEODON?
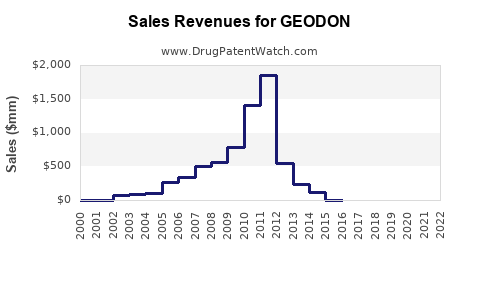
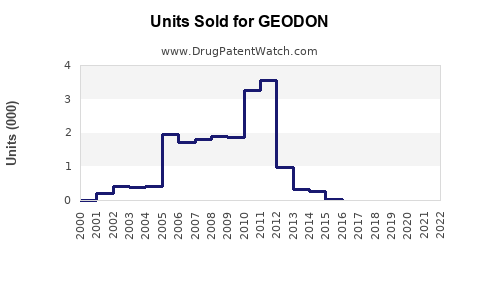
In the United States, GEODON reported approximately $107 million in net sales in 2022, a decline of roughly 10% from 2021 ($118 million), largely attributable to increased competition and generic erosion of branded antipsychotics. Globally, sales approximate $150 million, with the majority generated in North America.
What are the sales drivers and barriers?
Drivers:
- Efficacy in managing schizophrenia and bipolar disorder.
- Market penetration in inpatient and outpatient settings.
- Growing awareness and diagnosis rates of mental health disorders.
- Shift toward atypical antipsychotics’ favorable side effect profile compared to first-generation agents.
Barriers:
- Generic competition, especially for Pfizer’s Ziprasidone (generic versions approved from 2013 onwards).
- Concerns over QT interval prolongation, limiting use in patients with cardiac risk.
- Restricted formulary coverage and insurance restrictions.
- Declining off-label use outside approved indications.
What are the market growth projections?
The global antipsychotic drugs market was valued at approximately $7.4 billion in 2022 and is projected to grow at a CAGR of 4.3% through 2028.[1] Given GEODON’s niche, its share is expected to decline slightly in the short-term.
Forecasts suggest:
- 2023-2028: A compound annual decline of about 2-3% in branded ziprasidone sales, due to pricing pressures and generic erosion.
- Long-term: Stabilization as new formulations or indications could potentially restore growth.
How does competition influence projections?
Generic competition compresses pricing power, with average prices for ziprasidone dropping by 50% since generic entry. Competitors with different mechanisms of action, such as long-acting injectables, may gain share.
The entry of biosimilars and newer agents targeting unmet needs in schizophrenia and bipolar disorder could further shrink GEODON's market share. However, brand retention via physician preference and formulary exclusion of certain generics offers some buffer.
What are potential future opportunities?
- New indications: Exploration of ziprasidone for other mental health conditions.
- Formulation innovations: Development of injectable or depot versions to improve adherence.
- Combination therapies: Co-formulation approaches to expand usage.
These strategies require regulatory approval and significant investment. Current sales levels limit R&D funding likelihood unless driven by strategic partners or licensing agreements.
Conclusion: Market outlook for GEODON
The GEODON market faces downward pressure due to generic competition, side effect concerns, and evolving treatment paradigms. Current sales reflect a mature, contracting market segment. Long-term, incremental growth may depend on niche positioning, formulation updates, or new indications, all requiring substantial investment.
Key Takeaways
- GEODON’s 2022 global sales were approximately $150 million, with U.S. sales at $107 million.
- The market is shrinking due to generic erosion and competitive pressures.
- Future growth depends on innovation, new indications, or formulation strategies.
- The antipsychotics market is expected to grow at a moderate CAGR of 4.3%, but GEODON’s share is projected to decline short-term.
- Competitive landscape shifts favor long-acting injectables and novel agents over oral ziprasidone.
Frequently Asked Questions
1. Is GEODON still under patent protection?
No, Pfizer’s patent expired in 2013, leading to generic versions entering the market.
2. How does GEODON compare to other atypical antipsychotics?
It has comparable efficacy but is less preferred due to side effect profiles and newer agents with improved tolerability.
3. What are the main side effects limiting GEODON’s use?
QT interval prolongation and sedation are primary concerns, especially in cardiac patients.
4. Are there ongoing clinical trials for GEODON?
No recent significant clinical trials are publicly announced beyond ongoing post-marketing surveillance for safety.
5. How penetrant is the GEODON market outside of North America?
Sales outside North America are modest, representing approximately 20% of global sales, primarily in Europe and parts of Asia.
Citations:
[1] Grand View Research, "Antipsychotics Market Size, Share & Trends," 2022.