Last updated: February 13, 2026
What Is the Current Market Size and Distribution for Fluoxetine?
Fluoxetine, marketed primarily as Prozac, is a first-generation SSRI approved in 1987 for depression, OCD, bulimia, and panic disorder. The global antidepressant market was valued at approximately $19 billion in 2022, with fluoxetine accounting for an estimated 15-20% of sales, reflecting its historical prevalence and established market presence.
In 2022, the global fluoxetine market's value was roughly $3 billion. The United States accounts for about 45% of this, driven by high prescription rates and favorable insurance coverage. Europe contributes approximately 30%, with the remaining 25% distributed across Asia-Pacific, Latin America, and other regions.
Market share is relatively stable, with generic formulations accounting for over 85% of sales, while branded sales (e.g., Prozac) decline due to patent expirations and price competition. The rise of new antidepressants, especially SNRIs and novel therapeutics, has slightly constrained growth but not led to significant market share loss for fluoxetine.
What Are the Key Drivers and Challenges Affecting Future Sales?
Drivers
- Extensive clinical approval and long-term safety record foster continued prescribing.
- Generic availability reduces costs and widens access.
- Recent research explores fluoxetine’s potential for additional indications, such as obsessive-compulsive spectrum disorders, increasing off-label use.
- Growing mental health awareness and destigmatization expand treatment Uptake, especially post-pandemic.
Challenges
- Emergence of newer antidepressants with more rapid onset, fewer side effects, and broader mechanisms of action threaten market share.
- Regulatory restrictions in certain regions limit off-label uses.
- Potential safety concerns associated with long-term SSRI use, such as increased risk of suicidal ideation among adolescents, influence prescribing behavior.
- Patent expirations have led to price erosion, impacting profit margins.
What Are Recent Sales Trends and Growth Projections?
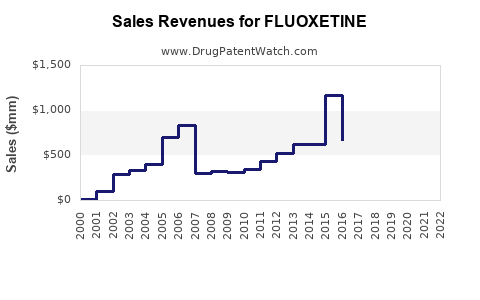
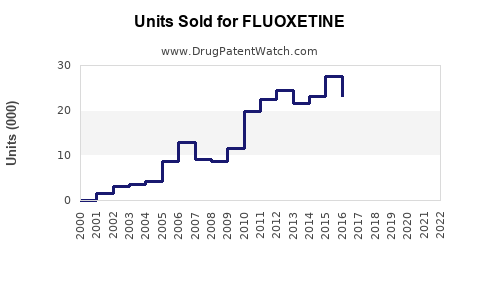
Sales peaked in the early 2000s at approximately $3.5 billion globally, driven by high initial brand sales. Since then, sales have plateaued and declined modestly due to generic competition, with an annual decrease of about 2-4%.
Projected Market Trends (2023-2027)
- The global fluoxetine market is forecasted to stabilize around $2.8 billion by 2027.
- CAGR projected at roughly -1% to 0%, reflecting market saturation, but with regions like Asia-Pacific experiencing modest growth due to increasing mental health services.
- In the U.S., prescription volumes for fluoxetine are expected to decline marginally due to shifting prescriber preferences toward newer agents but will remain significant because of entrenched usage patterns.
Sales by Region (2022)
| Region |
Market Share |
Estimated Sales ($ millions) |
| United States |
45% |
$1.35 billion |
| Europe |
30% |
$900 million |
| Asia-Pacific |
15% |
$450 million |
| Latin America & Others |
10% |
$300 million |
How Do Patent and Regulatory Statuses Influence Sales?
Fluoxetine's primary patent protections expired in the early to mid-2000s, leading to a surge in generics. This eroded branded sales, which now comprise less than 15% of total revenue. Regulatory agencies have approved multiple formulations, including once-daily and liquid forms, broadening access but also increasing competition.
The primary branded drug once held exclusivity until 2001 in the U.S., after which generics took over. Recent patent litigation and market entries are unlikely to significantly impact sales further, given the mature status of the drug.
What Are Competitive Dynamics and Potential Entrants?
Key competitors include newer SSRIs (sertraline, citalopram), SNRIs (venlafaxine, duloxetine), and emerging drugs targeting novel pathways such as glutamate or neurotrophic factors.
Market entry of biosimilars or combination therapies is negligible due to chemical small-molecule nature of fluoxetine.
The generic landscape is highly saturated, discouraging new entrants. Market dynamics favor price competition rather than innovation, limiting sales growth prospects.
What Are Future Opportunities and Risks?
Opportunities:
- Repurposing fluoxetine for off-label indications like premenstrual dysphoric disorder or post-traumatic stress disorder might open niche markets.
- Digitally integrated treatment plans could increase adherence and prescription frequency.
Risks:
- Regulatory shifts addressing SSRI safety concerns.
- Adoption of alternative therapies with better side-effect profiles.
- Emerging personalized medicine approaches favoring individualized treatment pathways over broad-spectrum drugs like fluoxetine.
Key Takeaways
- The fluorinated serotonin reuptake inhibitor market comprises approximately $3 billion globally, with fluoxetine comprising a significant but declining share.
- The drug's mature patent status and extensive generic competition limit growth, projected to decline modestly over the next five years.
- Regional differences influence market dynamics, with the U.S. maintaining the largest share.
- Brand sales historically peaked in the early 2000s, with steady erosion since due to generics.
- Off-label uses, safety profiles, and competition from newer drugs define future market potential.
FAQs
1. Will fluoxetine regain market share with new formulations or indications?
No significant resurgence is anticipated. Market saturation and competition from newer drugs limit growth opportunities. Off-label and niche use expansions are unlikely to offset overall decline.
2. How are regulatory changes affecting fluoxetine sales?
Stricter safety regulations and labeling requirements may reduce prescribing in some groups. However, no recent restrictive policies have notably curtailed existing prescription volumes.
3. What regional markets are emerging as growth opportunities?
Asia-Pacific shows modest growth due to increasing mental health awareness and expanding healthcare infrastructure, but overall impact remains limited by existing generics.
4. How significant is off-label prescribing for fluoxetine?
Off-label use accounts for an estimated 10-15% of prescriptions, primarily for conditions like premature ejaculation and certain eating disorders, potentially sustaining some sales.
5. Are biosimilars or novel formulations expected to influence the market?
No. Since fluoxetine is a chemical small molecule, biosimilar development is unnecessary; reformulations are primarily generic equivalents without significant market disruption.
References
- GlobalData. "Antidepressants Market Report 2022."
- IMS Health. "Prescriptions Data 2022."
- U.S. Food & Drug Administration. "Drug Approvals and Patent Status."
- Statista. "Market Size of Antidepressants 2022."
- MarketWatch. "Fluoxetine Market Analysis and Forecast 2023-2027."