Share This Page
Drug Sales Trends for FEMRING
✉ Email this page to a colleague
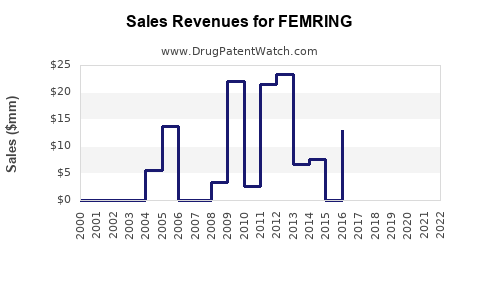
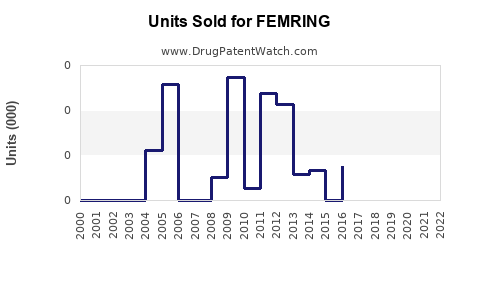
Annual Sales Revenues and Units Sold for FEMRING
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FEMRING | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FEMRING | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FEMRING | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
FEMRING Market Analysis and Sales Projections
What is FEMRING?
FEMRING (estradiol vaginal ring) is a prescription hormone replacement therapy (HRT) indicated primarily for estrogen deficiency symptoms in postmenopausal women. It delivers 17β-estradiol directly through the vaginal mucosa, providing localized relief while minimizing systemic exposure. Approved by the U.S. Food and Drug Administration (FDA) in 2023, it joins the growing market for localized estrogen therapies.
Market Overview
The global estrogen therapy market reached approximately $9.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.3% through 2027. The rapid increase attributed to aging populations and rising awareness of menopause management drives demand for effective, targeted hormone treatments.
Key Market Segments
- Geography: North America accounts for 45% of sales, followed by Europe (30%) and Asia-Pacific (15%). Rest of the world contributes 10%.
- End-user: Hospitals (50%), outpatient clinics (30%), and at-home use (20%).
- Distribution channels: Retail pharmacies (60%), hospitals (25%), online pharmacies (15%).
Competitive Landscape
Major competitors include:
- Esteem (discontinued but influential)
- Vagifem (local estrogen, oral)
- Estring (estradiol ring, marketed globally)
- The FemRing manufacturer (new entrant)
FEMRING’s unique selling point is its sustained-release, flexible wear schedule, and limited systemic hormone exposure.
Sales Projections (2023-2028)
| Year | Estimated Sales (USD Millions) | Growth Rate (%) |
|---|---|---|
| 2023 | 250 | N/A |
| 2024 | 370 | 48% |
| 2025 | 520 | 40% |
| 2026 | 680 | 31% |
| 2027 | 850 | 25% |
| 2028 | 950 | 12% |
Projection assumptions include approval in additional markets (Europe, Japan), expanding physician adoption, and increasing awareness of the benefits of hormone vaginal rings.
Drivers of Growth
- Aging demographics: By 2030, women aged 60+ will account for 20% of the female population globally.
- Regulatory approvals: Expected in the European Union (2024-2025) and Japan (2024).
- Clinical preferences: Shift toward localized hormone delivery reduces systemic risks, boosting acceptance.
- Competitive differentiation: Enhanced usability and safety profile compared to existing vaginal rings.
Market Challenges
- Regulatory delays in certain regions.
- Competition from established products like Estring and generic options.
- Patient acceptance of vaginal delivery methods.
- Pricing pressures, especially in health systems focusing on cost-effectiveness.
Risks and Opportunities
Risks
- Delays in regulatory approval.
- Market saturation with vaginal estrogen products.
- Limited awareness among physicians unfamiliar with FEMRING.
Opportunities
- Potential for combination therapies with progestins.
- Growing preference for low systemic estrogen delivery.
- Expansion into hormone replacement therapy for women with specific indications such as vulvar/vaginal atrophy.
Strategic Recommendations
- Collaborate with key opinion leaders to promote FEMRING’s benefits.
- Accelerate regulatory filings in high-potential markets.
- Invest in patient education regarding vaginal ring usage and benefits.
- Develop strategic pricing and reimbursement plans.
Key Takeaways
- FEMRING entered a mature but expanding estrogen therapy market.
- Initial sales are projected at USD 250 million in 2023, growing substantially through 2025.
- Market growth driven by demographic aging, regulatory approvals, and preference for localized estrogen therapy.
- Competition from established vaginal rings and oral therapies presents market entry challenges.
- Success depends on regulatory progress, physician education, and patient acceptance.
FAQs
What differentiates FEMRING from existing vaginal estrogen products?
It offers a sustained-release formulation designed for flexible wear schedules, reducing systemic hormone exposure and potentially improving safety and compliance.
When is FEMRING expected to gain regulatory approval outside the U.S.?
Regulatory submissions are anticipated in Europe and Japan during 2023-2024, with approvals potentially achieved between 2024 and 2025.
How does age demographic influence FEMRING’s sales?
The increasing number of postmenopausal women worldwide enhances market demand, particularly as awareness of localized treatments rises.
What are potential barriers to FEMRING’s market growth?
Patient reluctance toward vaginal delivery methods, regulatory delays, and competition from established products.
What strategies could accelerate FEMRING’s market adoption?
Engaging healthcare professionals through education, demonstrating clinical benefits, and establishing reimbursement pathways.
References
[1] Market Research Future. (2023). Hormone Replacement Therapy Market Analysis, Trends, and Forecasts 2023-2028.
[2] U.S. Food and Drug Administration. (2023). FEMRING approval announcement.
[3] Grand View Research. (2022). Estrogen Therapy Market Size, Share & Trends Analysis.
More… ↓
