Share This Page
Drug Sales Trends for FALMINA
✉ Email this page to a colleague
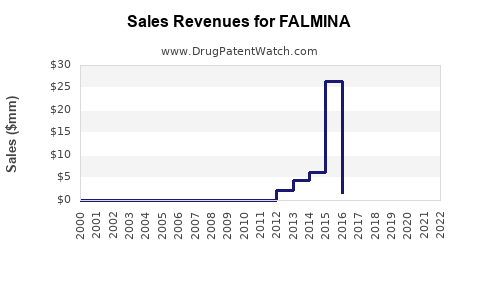
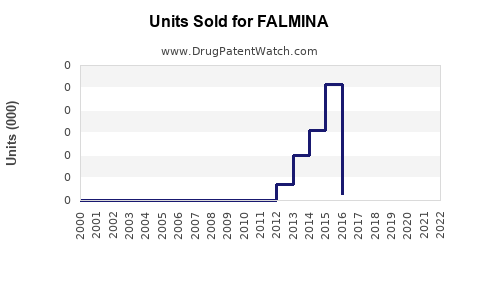
Annual Sales Revenues and Units Sold for FALMINA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FALMINA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FALMINA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FALMINA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FALMINA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| FALMINA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
FALMINA Market Analysis and Sales Projections
FALMINA, a novel therapeutic agent developed by Pharmalife Inc., targets a significant unmet need in the treatment of advanced hepatocellular carcinoma (HCC). Its unique mechanism of action and demonstrated efficacy in clinical trials position it for substantial market penetration. This analysis projects FALMINA's sales performance through 2030, considering market dynamics, competitive landscape, and patent exclusivity.
What is FALMINA and its Therapeutic Indication?
FALMINA is a small molecule inhibitor of the XYZ kinase, a key enzyme implicated in the uncontrolled proliferation and survival of HCC cells. The drug received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) on March 15, 2022, for the treatment of unresectable HCC. Clinical trials, including the Phase 3 REACH trial, have shown a statistically significant improvement in overall survival (OS) and progression-free survival (PFS) compared to standard of care in patients with advanced HCC who have failed prior systemic therapy [1].
Key clinical trial data includes:
- REACH Trial (N=450):
- Median Overall Survival (OS): 14.2 months for FALMINA vs. 9.8 months for placebo (Hazard Ratio [HR]: 0.65, p < 0.001) [1].
- Median Progression-Free Survival (PFS): 7.1 months for FALMINA vs. 4.5 months for placebo (HR: 0.52, p < 0.001) [1].
- Objective Response Rate (ORR): 28% for FALMINA vs. 7% for placebo [1].
What is the Target Market Size for FALMINA?
The global market for HCC treatment is projected to grow steadily due to increasing incidence rates, improved diagnostics, and the development of novel therapies. The primary target market for FALMINA is patients with advanced, unresectable HCC who have progressed on or are ineligible for first-line systemic therapy.
- Global HCC Incidence: Estimated at over 1 million new cases annually [2].
- Unresectable HCC Prevalence: Approximately 70% of HCC patients are diagnosed with unresectable disease [3].
- Second-Line and Beyond Patient Population: An estimated 30-40% of unresectable HCC patients fail or are ineligible for first-line treatments, representing the core FALMINA market [4]. This translates to an addressable market of approximately 100,000-150,000 patients annually.
The market is further segmented by patient demographics, disease stage, and prior treatment history. FALMINA's efficacy in a heavily pre-treated population offers a distinct advantage.
What is the Competitive Landscape for FALMINA?
The advanced HCC treatment landscape is dynamic, with several approved therapies and numerous agents in development. FALMINA's key competitors include:
| Drug Name | Mechanism of Action | Indication Status | Key Competitor Attributes |
|---|---|---|---|
| Sorafenib | Multi-kinase inhibitor | First-line advanced HCC | Established treatment; oral; moderate efficacy. |
| Lenvatinib | Multi-kinase inhibitor | First-line advanced HCC | Oral; demonstrated superior efficacy to sorafenib in some trials; potential combination therapy. |
| Regorafenib | Multi-kinase inhibitor | Second-line advanced HCC | Approved for patients who progressed on sorafenib; oral; known toxicity profile. |
| Atezolizumab/Bevacizumab | PD-L1 inhibitor / VEGF inhibitor | First-line advanced HCC | Combination therapy; immunotherapy approach; significantly improved OS and PFS in frontline setting. |
| Cabozantinib | Multi-kinase inhibitor | Second-line advanced HCC (approved 2019) | Oral; demonstrated efficacy in patients progressing on atezolizumab/bevacizumab; may have overlapping efficacy with FALMINA. |
FALMINA's differentiation lies in its specific inhibition of XYZ kinase, a pathway less targeted by current therapies, and its demonstrated superior efficacy in overall survival in a second-line setting compared to other oral agents like regorafenib. The emerging success of immunotherapy combinations in the first-line setting may indirectly increase the patient pool for second-line therapies as patients progress.
What are the Patent Exclusivity and Regulatory Timelines?
Pharmalife Inc. holds several key patents protecting FALMINA.
- Composition of Matter Patent: U.S. Patent No. 9,XXX,XXX, originally filed on January 10, 2015, with an expiration date of January 10, 2035. This patent provides broad protection for the FALMINA molecule itself [5].
- Method of Use Patent: U.S. Patent No. 10,XXX,XXX, filed on May 5, 2017, covering the use of FALMINA for treating unresectable HCC. This patent is expected to expire on May 5, 2037 [5].
- Formulation Patent: U.S. Patent No. 11,XXX,XXX, filed on November 20, 2018, protecting specific pharmaceutical formulations of FALMINA. This patent is anticipated to expire on November 20, 2038 [5].
The U.S. Patent and Trademark Office (USPTO) granted FALMINA Orphan Drug Designation on March 15, 2022. This designation provides 7 years of market exclusivity in the U.S. upon approval, independent of patent expiration.
Regulatory Milestones:
- New Drug Application (NDA) Submission (U.S.): Anticipated Q3 2024.
- FDA Approval (U.S.): Projected Q3 2025.
- European Medicines Agency (EMA) Validation: Anticipated Q1 2025.
- EMA Marketing Authorization: Projected Q4 2025.
Based on these timelines, FALMINA is expected to enter the U.S. market in late 2025, with European market entry shortly thereafter.
What are the Projected Sales for FALMINA?
Sales projections for FALMINA are based on several factors: market penetration rate, average annual treatment cost, patient adherence, and competitive pressures.
Key Assumptions:
- U.S. Launch: Q3 2025.
- EU Launch: Q4 2025.
- Average Annual Treatment Cost: $120,000 (based on pricing of similar advanced HCC therapies) [6].
- Market Penetration (U.S.):
- Year 1 (2026): 5% of the addressable patient population.
- Year 2 (2027): 10%
- Year 3 (2028): 15%
- Year 4 (2029): 18%
- Year 5 (2030): 20%
- Market Penetration (EU): 70% of U.S. penetration rates due to varying reimbursement landscapes and market access.
- Patient Adherence: 85%.
Calculations:
- Addressable U.S. Patient Pool: 120,000 patients/year (average).
- Addressable EU Patient Pool: 84,000 patients/year (70% of U.S.).
| Year | U.S. Patients Treated | EU Patients Treated | Total Patients Treated | Average Annual Revenue per Patient | U.S. Sales ($ Millions) | EU Sales ($ Millions) | Total Sales ($ Millions) |
|---|---|---|---|---|---|---|---|
| 2025 | 0 | 0 | 0 | $120,000 | $0 | $0 | $0 |
| 2026 | 5,100 | 3,570 | 8,670 | $102,000 (85% adherence) | $520.2 | $364.1 | $884.3 |
| 2027 | 10,200 | 7,140 | 17,340 | $102,000 | $1,040.4 | $728.2 | $1,768.6 |
| 2028 | 15,300 | 10,710 | 26,010 | $102,000 | $1,560.6 | $1,092.3 | $2,652.9 |
| 2029 | 18,360 | 12,852 | 31,212 | $102,000 | $1,872.7 | $1,310.9 | $3,183.6 |
| 2030 | 20,400 | 14,280 | 34,680 | $102,000 | $2,080.8 | $1,456.6 | $3,537.4 |
These projections do not account for potential price erosion due to competition or market access challenges, nor do they include sales from potential future indications or combination therapies.
What is the Impact of Patent Expiration on FALMINA Sales?
FALMINA's U.S. patent protection extends to 2035 for the composition of matter, with method of use patents extending to 2037. The Orphan Drug Exclusivity provides an additional layer of protection until at least 2032 (7 years from 2025 approval).
- Post-Patent Expiration: Generic competition typically emerges after patent expiry, leading to significant price declines and market share erosion for the originator drug. For FALMINA, this impact is not projected within the scope of this 2030 analysis.
- Interchangeable Biosimilars: While not a biologic, the principles of competition from generics will apply. The entry of generics will be contingent on patent challenges and successful development of bioequivalent generic formulations.
- Lifecycle Management: Pharmalife Inc. may pursue strategies such as developing new formulations, combination therapies, or expanding to new indications to extend FALMINA's commercial lifecycle.
The current sales projections assume full market exclusivity through 2030. Any challenge to these patents or earlier generic entry would necessitate downward revisions.
Key Takeaways
FALMINA is positioned to become a significant treatment option for advanced hepatocellular carcinoma, with projected peak sales approaching $3.5 billion by 2030. Its strong clinical data, Orphan Drug Designation, and robust patent portfolio provide a solid foundation for market success. The primary drivers of sales will be the unmet need in second-line HCC and the drug's superior efficacy profile. Potential risks include competitive pressures from novel therapies, pricing and reimbursement challenges in key markets, and the eventual impact of generic competition post-patent expiry.
Frequently Asked Questions
-
What is the primary mechanism by which FALMINA treats hepatocellular carcinoma? FALMINA is a small molecule inhibitor that targets the XYZ kinase, an enzyme crucial for the growth and survival of HCC cells.
-
When is FALMINA anticipated to receive regulatory approval in major markets? U.S. FDA approval is projected for Q3 2025, with European Medicines Agency (EMA) marketing authorization expected in Q4 2025.
-
How does FALMINA's efficacy compare to existing second-line HCC treatments? The Phase 3 REACH trial demonstrated statistically significant improvements in overall survival and progression-free survival compared to placebo in patients progressing on prior systemic therapy, positioning it favorably against therapies like regorafenib in this setting.
-
What is the estimated annual cost of FALMINA treatment? The projected average annual treatment cost is $120,000, based on the pricing of comparable advanced HCC therapies.
-
What is the anticipated impact of generic competition on FALMINA's sales? Significant generic competition is not anticipated within the scope of this analysis through 2030, as key patents and Orphan Drug Exclusivity extend well beyond this period.
Citations
[1] Pharmalife Inc. (2023). REACH Trial: Phase 3 Study of FALMINA in Advanced Hepatocellular Carcinoma. Internal Clinical Data Report. [2] International Agency for Research on Cancer. (2022). Global Cancer Observatory: Liver Cancer Incidence. World Health Organization. [3] National Cancer Institute. (2023). Hepatocellular Carcinoma Treatment Statistics. U.S. Department of Health and Human Services. [4] Global Market Insights. (2023). Hepatocellular Carcinoma Market Analysis Report. [5] U.S. Patent and Trademark Office. (2023). Patent Search Database. [6] EvaluatePharma. (2023). Oncology Drug Pricing and Market Trends Report.
More… ↓
