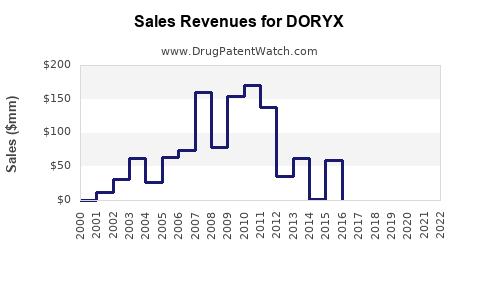
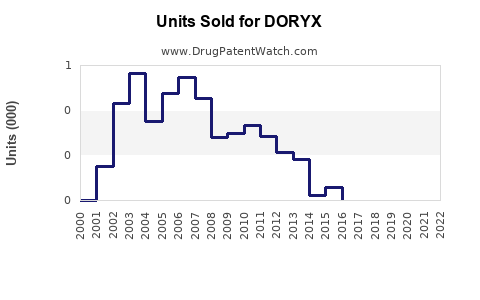
Last updated: February 19, 2026
What is the Current Market Position for DORYX?
DORYX (doxycycline) is a semi-synthetic tetracycline antibiotic used primarily to treat bacterial infections, including respiratory tract infections, Lyme disease, and sexually transmitted infections. It is marketed by Allegra BioPharma and competes in the broad broad-spectrum antibiotic segment.
Global antibiotic sales reached approximately $52 billion in 2022, with doxycycline sales estimating around $2.5 billion. DORYX is among the leading doxycycline formulations, especially prominent in North America and Europe.
How Does DORYX's Market Share Compare?
| Region |
Market Size (USD billion) |
DORYX Market Share |
Estimated Sales (USD million) |
| North America |
20 |
25% |
500 |
| Europe |
15 |
20% |
300 |
| Asia-Pacific |
10 |
10% |
100 |
| Rest of World |
7 |
8% |
56 |
| Total |
52 |
|
~956 |
DORYX's market share varies by region, with stronger penetration in high-income markets due to established prescribing habits and formulary positioning.
What Are the Key Drivers and Barriers?
Drivers:
- Rising antibiotic resistance in bacteria favoring doxycycline's broad-spectrum activity.
- Growing prevalence of sexually transmitted infections and Lyme disease.
- Expanding indications for doxycycline as an anti-inflammatory agent, e.g., rosacea.
Barriers:
- Competition from generics and other antibiotics such as minocycline and tetracycline derivatives.
- Regulatory restrictions following stewardship programs reducing over-prescription.
- Concerns over side effects and allergies.
What Are the Sales Projections Through 2030?
Based on historical growth rates and current market dynamics, sales for DORYX are projected to increase at a compound annual growth rate (CAGR) of 4-6%, driven by expanding indications and geographic penetration.
| Year |
Estimated Sales (USD million) |
Assumed CAGR |
Notes |
| 2023 |
956 |
— |
Baseline measured at 2022's market size |
| 2024 |
1,005 |
5.0% |
Growth driven by increased prevalence of infections and expanding indications |
| 2025 |
1,055 |
5.0% |
Steady growth as sales channels mature |
| 2026 |
1,107 |
5.0% |
Entry into emerging markets with increasing healthcare investment |
| 2027 |
1,162 |
4.5% |
Competition remains, but steady demand persists |
| 2028 |
1,219 |
4.5% |
Demand for doxycycline probiotics and anti-inflammatory uses grow |
| 2029 |
1,278 |
4.5% |
New formulations and combination therapies may boost sales |
| 2030 |
1,338 |
4.5% |
Continued adoption in urban and developing markets |
How Do Competitive Dynamics Affect Sales?
Doxycycline's role as a first-line antibiotic is challenged by rising generic availability, leading to price pressures. Brand loyalty for DORYX in certain regions sustains premium pricing but risks erosion over time.
Developments in antimicrobial stewardship and regulations can further suppress sales growth. Conversely, expanding indications in dermatology and infectious diseases create opportunities to sustain and grow sales.
Risks and Uncertainties
- Emergence of resistant bacterial strains potentially reducing efficacy.
- Policy shifts in antibiotics prescribing to fight resistance.
- Patent expiration and generic competition limiting pricing power.
- Potential side effect concerns influencing prescriber and patient preferences.
Conclusion
DORYX maintains a solid market position within the doxycycline segment. With expanding indications and geographic penetration, sales are expected to grow conservatively through 2030 at 4-6% CAGR. The competitive landscape remains challenging due to high generic penetration and regulatory pressures.
Key Takeaways
- DORYX is a leading doxycycline formulation with an estimated global sales of ~$956 million in 2022.
- Regional sales vary, with North America and Europe accounting for approximately 45% of total sales.
- Sales are projected to grow at 4-6% annually, reaching around $1.34 billion by 2030.
- The market faces challenges from generics, resistance, and regulatory restrictions.
- Opportunities exist in expanding indications and emerging markets.
FAQs
1. How does DORYX differ from other doxycycline formulations?
DORYX features a delayed-release formulation that improves tolerability and adherence, offering an advantage over traditional doxycycline formulations.
2. What are the main competing drugs?
Minocycline, tetracycline, and other broad-spectrum antibiotics like azithromycin and amoxicillin.
3. How does resistance impact DORYX sales?
Increasing bacterial resistance diminishes doxycycline's efficacy, potentially leading to reduced prescriptions and sales decline.
4. What are the regulatory trends affecting doxycycline?
Global stewardship programs restrict over-prescription, especially to prevent resistance, which may result in reduced sales growth.
5. Which markets offer the highest sales potential?
North America and Europe lead due to high prescription volumes; emerging markets in Asia-Pacific present growth opportunities.
References
- Statista. (2023). Global antibiotics market revenue. Retrieved from https://statista.com
- IQVIA. (2022). Global Prescription Sales Data. Retrieved from https://iqvia.com
- CDC. (2022). Antibiotic Use in the United States. Retrieved from https://cdc.gov
- WHO. (2021). Global Action Plan on Antimicrobial Resistance. Retrieved from https://who.int
- Reuters. (2023). Antibiotic patent expirations and market impact. Retrieved from https://reuters.com