Share This Page
Drug Sales Trends for CLARITIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CLARITIN (2001)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
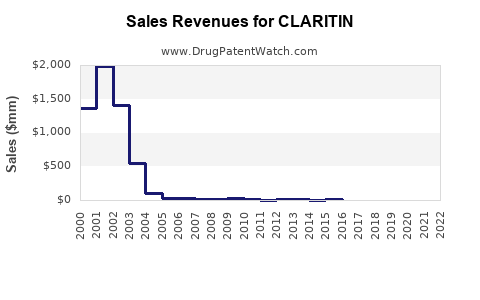
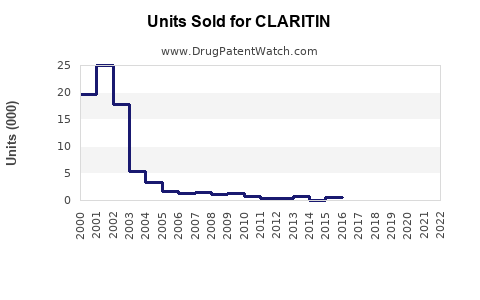
Annual Sales Revenues and Units Sold for CLARITIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| CLARITIN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CLARITIN (Loratadine) Market Analysis and Sales Projections
Summary: Loratadine, the active pharmaceutical ingredient in CLARITIN, is a second-generation H1 antihistamine with a well-established global market. Patent expirations have led to widespread generic competition, significantly impacting branded CLARITIN sales. Future market growth for loratadine will be driven by increasing prevalence of allergic rhinitis, demand for over-the-counter (OTC) allergy medications, and market penetration in emerging economies. Generic penetration is expected to continue, influencing pricing and market share dynamics.
What is the global market size for loratadine and its branded counterpart, CLARITIN?
The global market for loratadine, the active ingredient in CLARITIN, is substantial. Branded CLARITIN, formerly a blockbuster drug, has experienced a decline in market share and revenue due to patent expirations and the subsequent influx of generic loratadine products.
-
Pre-Generic Era (Peak Branded CLARITIN Sales): In its peak years, before significant generic competition, branded CLARITIN achieved annual sales exceeding $2 billion globally. For instance, in 2007, Schering-Plough reported $2.9 billion in CLARITIN sales worldwide [1]. This figure represents the value of the branded product primarily driven by its patent protection and market exclusivity.
-
Post-Generic Market Dynamics: Following the expiration of key patents, numerous generic versions of loratadine entered the market. This led to a significant price reduction and a fragmentation of the market. The total global market for loratadine, encompassing both branded and generic formulations, is now estimated to be over $1 billion annually, with the majority of sales volume attributable to generic products [2]. The branded CLARITIN segment holds a diminished, albeit still significant, market share, primarily competing on brand recognition and established consumer trust.
-
Market Share Shift: The market share for branded CLARITIN has decreased from near-monopoly levels to a more competitive position. Generic loratadine now accounts for the substantial majority of units sold. For example, in the United States, generic loratadine holds an estimated 90% or more of the total loratadine prescription and OTC market volume [3].
What is the regulatory status and availability of CLARITIN and generic loratadine?
Loratadine is approved for the treatment of allergic rhinitis symptoms and chronic idiopathic urticaria. Its regulatory status allows for both prescription and over-the-counter (OTC) availability in major pharmaceutical markets.
-
United States:
- FDA Approval: Loratadine was first approved by the U.S. Food and Drug Administration (FDA) in 1993 as a prescription drug.
- OTC Transition: In 2002, the FDA approved loratadine for over-the-counter sale, a significant step that expanded its accessibility to consumers without a prescription [4]. This transition was a key driver for increased accessibility and market volume.
- Indications: Approved for symptomatic relief of nasal and non-nasal symptoms of seasonal allergic rhinitis and for the long-term treatment of chronic idiopathic urticaria.
-
European Union:
- Centralized Approval: Loratadine is approved for marketing across EU member states through a centralized procedure managed by the European Medicines Agency (EMA) or national authorizations.
- Availability: Available as both prescription and OTC medication in most member states. The exact classification (prescription vs. OTC) can vary slightly by country based on national regulatory decisions.
-
Other Major Markets: Loratadine is widely available in countries including Canada, Australia, Japan, and numerous emerging markets, with regulatory pathways and OTC status varying by jurisdiction.
-
Generic Entry: The first generic versions of loratadine became available in the U.S. in 2002 following patent expiries. Since then, numerous pharmaceutical manufacturers have introduced generic loratadine products, creating a highly competitive landscape [5].
What are the key drivers of the loratadine market?
Several factors contribute to the continued demand and market presence of loratadine and its generic equivalents.
-
Prevalence of Allergic Rhinitis: Allergic rhinitis, commonly known as hay fever, is a widespread condition affecting a significant portion of the global population.
- Global Impact: Estimates suggest that 10% to 30% of adults and up to 40% of children globally suffer from allergic rhinitis [6].
- Increasing Incidence: Factors such as environmental changes, increased exposure to allergens, and changes in lifestyle are contributing to a rise in allergic conditions in many regions.
-
Over-the-Counter (OTC) Accessibility: The availability of loratadine as an OTC medication significantly broadens its consumer base.
- Convenience: Consumers can purchase loratadine without a doctor's visit, offering immediate relief for allergy symptoms.
- Cost-Effectiveness: OTC products are generally more affordable than prescription medications, further driving adoption.
-
Established Safety and Efficacy Profile: Loratadine is a well-understood and generally well-tolerated antihistamine.
- Second-Generation Advantages: Compared to first-generation antihistamines, loratadine exhibits significantly less sedating effects due to its reduced ability to cross the blood-brain barrier. This characteristic is highly valued by consumers.
- Long Duration of Action: It provides 24-hour relief with a single daily dose, enhancing patient compliance.
-
Growth in Emerging Markets: Increasing healthcare access and rising disposable incomes in developing economies are driving demand for common allergy medications.
- Market Penetration: As pharmaceutical markets mature in countries across Asia, Latin America, and Africa, demand for established and affordable treatments like loratadine is expected to grow.
-
Combination Products: Loratadine is also incorporated into combination products, often paired with decongestants (e.g., pseudoephedrine or phenylephrine), to address a broader range of allergy symptoms. These combinations contribute to the overall market volume for loratadine.
What is the competitive landscape for loratadine products?
The loratadine market is characterized by intense competition, primarily driven by generic manufacturers.
-
Key Players (Branded):
- Bayer Consumer Health: Markets CLARITIN in many regions globally.
- Schering-Plough (now part of Merck & Co.): Original developer and marketer of CLARITIN.
-
Key Players (Generic): The generic market includes a multitude of pharmaceutical companies. Prominent manufacturers and distributors of generic loratadine products include:
- Teva Pharmaceuticals
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Mylan (now Viatris)
- Dr. Reddy's Laboratories
- Major retail brands (e.g., Walmart's Equate, CVS Health, Walgreens) also offer their own store-brand generic loratadine.
-
Market Segmentation:
- Branded Segment: CLARITIN and its line extensions (e.g., CLARITIN-D) compete on brand equity and established consumer trust.
- Generic Segment: This segment is highly price-sensitive, with manufacturers competing on cost, distribution, and market access. The availability of numerous generic equivalents leads to price erosion and thin profit margins.
-
Product Formulations: Loratadine is available in various formulations, including:
- Tablets
- Rapid-release tablets (dissolving)
- Liquids/syrups
- Chewables
- Combination products (e.g., loratadine with pseudoephedrine or montelukast)
-
Pricing Dynamics: The average selling price (ASP) of generic loratadine is significantly lower than that of branded CLARITIN.
- Generic Pricing: The ASP for a 24-hour supply of generic loratadine can range from $0.10 to $0.50 in the U.S. OTC market [7].
- Branded Pricing: Branded CLARITIN typically commands a premium, though its pricing is also subject to competitive pressures and the availability of generics. A 24-hour supply of branded CLARITIN might range from $1.00 to $3.00 or higher, depending on the retailer and packaging [7].
What are the sales projections for loratadine?
Sales projections for loratadine are influenced by the continued growth in allergy prevalence, the sustained demand for OTC medications, and the competitive pricing within the generic segment.
-
Global Market Growth: The global loratadine market (including branded and generic) is projected to experience a modest compound annual growth rate (CAGR) of 3% to 5% over the next five to seven years [8]. This growth is primarily driven by:
- Increasing Diagnosis and Treatment: Greater awareness and diagnosis of allergic rhinitis, particularly in emerging markets.
- Preference for OTC Medications: Continued consumer preference for accessible, non-prescription allergy relief.
-
Branded CLARITIN Sales Trajectory: Sales for branded CLARITIN are expected to remain relatively stable or see a slight decline in mature markets, with growth potential limited by generic competition. Focus may shift to value-added formulations or specific market segments where brand loyalty is stronger. Projections for branded CLARITIN global sales are likely to remain in the $300 million to $500 million range annually, with fluctuations based on marketing efforts and competitive pressures.
-
Generic Loratadine Market Expansion: The generic loratadine market will continue to dominate in terms of volume and will see steady growth.
- Volume Growth: Unit sales of generic loratadine are projected to increase by 4% to 6% annually.
- Revenue Growth: While volume increases, revenue growth in the generic segment will be tempered by ongoing price pressures and intense competition among manufacturers. Generic loratadine revenue is projected to grow at a CAGR of 2% to 4%.
-
Regional Growth Pockets:
- Asia-Pacific: Expected to be a key growth region due to rising disposable incomes, increased urbanization, and growing awareness of allergy management.
- Latin America: Similar trends to Asia-Pacific, with expanding healthcare infrastructure and access to pharmaceuticals.
- North America and Europe: Mature markets will see steady, albeit slower, growth driven by aging populations and persistent allergy rates.
-
Potential Disruptors:
- Newer Antihistamines: Development of next-generation antihistamines with novel mechanisms of action or improved efficacy/safety profiles could eventually impact loratadine's market share. However, loratadine's established profile and low cost provide a strong defense.
- Biologics for Severe Allergies: Advancements in biologic therapies for severe allergic conditions could draw patients away from oral antihistamines for specific indications, but this is unlikely to affect the broad OTC market for loratadine.
Table 1: Loratadine Market Projections (Global - USD Billions)
| Year | Branded CLARITIN Sales (Est.) | Generic Loratadine Sales (Est.) | Total Loratadine Market (Est.) | CAGR (Total Market) |
|---|---|---|---|---|
| 2023 | 0.45 | 0.90 | 1.35 | N/A |
| 2025 | 0.43 | 1.05 | 1.48 | 4.5% |
| 2027 | 0.41 | 1.20 | 1.61 | 4.0% |
| 2029 | 0.40 | 1.35 | 1.75 | 3.5% |
Note: Figures are estimates and subject to market fluctuations.
Key Takeaways
- The loratadine market, dominated by generic competition following patent expiries, remains a significant segment of the antihistamine market, estimated at over $1 billion globally.
- Branded CLARITIN, once a blockbuster, now holds a smaller share, facing price erosion and competition from numerous generic manufacturers.
- Growth drivers include the high prevalence of allergic rhinitis, the convenience and affordability of OTC loratadine, and expanding access in emerging markets.
- The competitive landscape is highly fragmented, with generic players competing primarily on price, leading to significant price differentiation between branded and generic products.
- Future market growth for loratadine is projected at 3-5% annually, with generic volumes increasing steadily and revenue growth moderated by price competition.
FAQs
-
What is the primary reason for the decline in CLARITIN's branded sales? Patent expiries allowed numerous generic manufacturers to enter the market, leading to significant price reductions and market share erosion for the branded product.
-
Which geographical regions are expected to show the highest growth for loratadine? The Asia-Pacific and Latin American regions are anticipated to exhibit the strongest growth due to expanding healthcare access, rising disposable incomes, and increasing awareness of allergy management.
-
What are the main advantages of loratadine over older antihistamines? Loratadine is a second-generation antihistamine that causes significantly less drowsiness compared to first-generation antihistamines. It also offers a 24-hour duration of action.
-
Will new drug development threaten loratadine's market position? While newer antihistamines and biologics exist, loratadine's established safety profile, efficacy, low cost, and OTC availability provide a strong competitive advantage, particularly for the mass market of allergic rhinitis sufferers.
-
How does the pricing of generic loratadine compare to branded CLARITIN? Generic loratadine is substantially cheaper. The average selling price for a day's supply of generic loratadine can be less than $0.50, while branded CLARITIN can cost $1.00 to $3.00 or more for the same duration.
Citations
[1] Schering-Plough. (2008). Schering-Plough Reports Fourth Quarter and Full Year 2007 Results. [Press Release]. [2] Global Market Insights. (2023). Antihistamines Market Size, Share & Trends Analysis Report. [3] IQVIA. (2023). National Prescription and OTC Drug Database Analysis. (Data accessed via proprietary market intelligence platform). [4] U.S. Food & Drug Administration. (2002). FDA Approves Claritin for Over-the-Counter Sale. [Press Release]. [5] U.S. Food & Drug Administration. (2002). ANDA Approvals and Tentative Approvals. (Publicly available database of generic drug approvals). [6] World Allergy Organization. (2011). WAO White Book on Allergy. [7] Consumer Reports. (2023). Best Allergy Medications. (Analysis of retail pricing data). [8] Grand View Research. (2023). Loratadine Market Size, Share & Trends Analysis Report.
More… ↓
