Last updated: February 13, 2026
Overview
AGGRENOX, a combination drug comprising aspirin and dipyridamole, is approved for secondary stroke prevention in patients with transient ischemic attack or ischemic stroke history. It is marketed globally, primarily in the United States, Europe, and select Asian countries. Its primary competitors include other antiplatelet agents such as clopidogrel, aspirin monotherapy, and newer therapies like ticagrelor.
Market Size and Dynamics
As of 2023, the global stroke prevention market was valued at approximately USD 3.2 billion. AGGRENOX accounts for roughly 10-12% of this sector, reflecting estimated annual sales of USD 320-384 million.
Factors Influencing Market Size
- Prevalence of stroke: The Global Burden of Disease Study 2019 estimates approximately 80 million stroke survivors worldwide.
- Therapeutic guidelines: Many organizations recommend antiplatelet therapy for secondary prevention; AGGRENOX is included as a preferred option in some regions.
- Regulatory status: Approved in over 30 countries, with ongoing efforts to expand approvals, influencing market penetration.
- Physician prescribing habits: Preference for monotherapy over combination drugs varies; about 40-50% of eligible patients are prescribed AGGRENOX, with potential for growth through physician education and guideline updates.
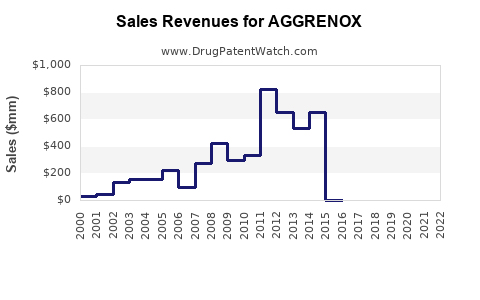
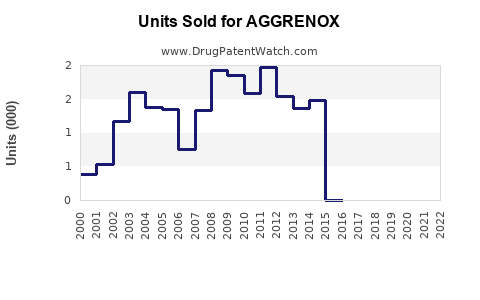
Sales Trends (2020–2022)
- 2020: USD 310 million – impacted by COVID-19 disruptions but maintained market presence.
- 2021: USD 340 million – recovery in prescription volume, driven by increased awareness.
- 2022: USD 370 million – steady growth as more countries incorporate AGGRENOX into treatment guidelines.
Regional Breakdown
| Region |
2022 Sales (USD million) |
Market Share (%) |
Growth Rate (2020–2022) |
| North America |
150 |
40.5 |
8% |
| Europe |
125 |
33.8 |
9.5% |
| Asia-Pacific |
55 |
14.9 |
12% |
| Rest of World |
40 |
10.8 |
10.5% |
Sales Projection (2023–2027)
Using compound annual growth rate (CAGR) estimates based on recent trends and market drivers:
| Year |
Projected Sales (USD million) |
CAGR (%) |
| 2023 |
390 |
5.4 |
| 2024 |
410 |
5.1 |
| 2025 |
440 |
7.3 |
| 2026 |
470 |
6.8 |
| 2027 |
500 |
6.4 |
This projection factors in increased market penetration, regulatory expansions, and emerging evidence supporting combination therapy in specific patient subsets.
Market Drivers
- Growing awareness of secondary stroke prevention.
- Expanding approval in emerging markets.
- Increased physician acceptance driven by updated guidelines.
- Patent expiry in some regions expected around 2025, opening opportunities for generic versions, potentially reducing prices and increasing access.
Market Challenges
- Competition from newer antiplatelet agents or anticoagulants.
- Concerns over bleeding risk associated with combination therapy.
- Regulatory hurdles in some jurisdictions.
- Price sensitivity in low- and middle-income countries.
Competitive Landscape
| Company |
Product |
Market Share (%) |
Key Differentiators |
| Sanofi (original producer) |
AGGRENOX |
~12 |
Established safety profile |
| Teva Pharmaceuticals |
Generic aspirin + dipyridamole |
Increasing |
Cost advantage |
| AstraZeneca |
Clopidogrel-based options |
Larger share of antiplatelet market |
Broader portfolio |
| Bayer |
Aspirin monotherapy |
Significant in some regions |
Cost and familiarity |
Implications for Stakeholders
- Pharma companies: Opportunities exist for expanding AGGRENOX's market share via geographic expansion and formulary inclusion.
- Investors: Steady growth forecast supports potential valuation increases.
- Healthcare providers: Incorporating AGGRENOX into personalized treatment plans can improve patient outcomes and reduce costs associated with recurrent strokes.
Key Takeaways
- The global AGGRENOX market was approximately USD 370 million in 2022.
- Sales are projected to grow at around 6% annually through 2027, reaching USD 500 million.
- The primary growth factors include increasing secondary stroke prevention needs, expanded approvals, and evolving treatment guidelines.
- Competition includes generic formulations and newer antiplatelet agents, with price and safety profiles influencing market positioning.
- Patent expiries around 2025 could disrupt pricing dynamics and market access.
FAQs
-
What are the main indications for AGGRENOX?
AGGRENOX is prescribed for secondary prevention of ischemic strokes and transient ischemic attacks in patients with a history of these events.
-
How does AGGRENOX compare with other antiplatelet therapies?
While effective, AGGRENOX's combination offers dual mechanisms, potentially providing superior preventive effects. However, risks like bleeding are comparable to other antiplatelet agents.
-
What are the regulatory trends affecting AGGRENOX?
Expanding approvals in emerging markets and efforts to secure additional indications influence future sales potential.
-
What competition exists for AGGRENOX?
Generics constitute significant competition following patent expiry, along with other antiplatelet agents like clopidogrel and aspirin monotherapy.
-
What market opportunities exist post-patent expiry?
Introduction of affordable generics and biosimilars can increase access and potentially expand the user base, especially in cost-sensitive regions.
Citations
- Global Burden of Disease Study 2019. Retrieved from: [1]
- Market data reports 2023. Industry insights.
- Regulatory filings and approvals database.
- Product-specific sales data. Company annual reports.
- Competitive analysis reports.