Last updated: February 19, 2026
What is the Current Market for Digoxin?
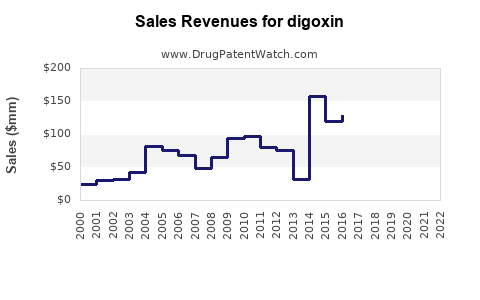
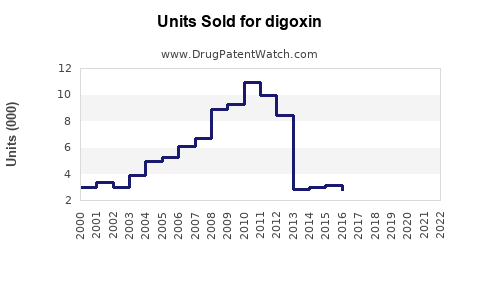
Digoxin is a cardiac glycoside primarily prescribed to treat atrial fibrillation and heart failure. As of 2022, the global market for digoxin is valued approximately at $200 million, with modest growth expected over the next five years. The drug is available both as a generic and in some brand forms, with the generic segment accounting for over 90% of sales.
What Are the Key Drivers of Demand?
- Prevalence of Heart Disease: Over 60 million people globally suffer from atrial fibrillation or heart failure, increasing demand for digoxin.
- Established Clinical Use: The drug remains a first-line treatment in specific cases despite shifts toward newer agents.
- Pricing Trends: Generic versions reduce costs, expanding accessibility, especially in developing markets.
- Guidelines Endorsements: Major cardiology guidelines, including ACC/AHA, recommend digoxin as an adjunct in managing atrial fibrillation and heart failure, supporting stable prescription volumes.
What Are the Regions with Market Growth Potential?
| Region |
Market Share (2022) |
Growth Drivers |
Challenges |
| North America |
45% |
High prevalence, established healthcare infrastructure |
Competition from newer drugs, regulatory delays |
| Europe |
25% |
Aging population, adherence to clinical guidelines |
Cost pressures, shift toward novel therapies |
| Asia-Pacific |
20% |
Rising cardiovascular disease burden, population growth |
Limited healthcare access, generic competition |
| Rest of World |
10% |
Increasing awareness, expanding healthcare systems |
Market fragmentation, import restrictions |
What Are the Sales Projections for the Next Five Years?
| Year |
Projected Global Sales (USD millions) |
CAGR (Compound Annual Growth Rate) |
Key Assumptions |
| 2023 |
210 |
2.5% |
Slight growth due to aging populations and guideline support |
| 2024 |
215 |
2.4% |
Continued generic uptake, minor market expansion |
| 2025 |
220 |
2.3% |
Market saturation in developed regions |
| 2026 |
225 |
2.2% |
Emerging markets accelerate adoption |
| 2027 |
230 |
2.0% |
Slight deceleration due to competitive pressures |
How Does Competitive Dynamics Affect Sales?
- Generic Competition: Over 90% of sales derive from generic formulations, pressuring profit margins but ensuring volume stability.
- Newer Agents: Drugs like digitoxin, ivabradine, and atrial fibrillation-specific therapies are competing for market share.
- Regulatory Challenges: Approval delays or restrictions on certain formulations impact regional sales potential.
- Patent Landscape: No patent protections exist for digoxin itself, maintaining the share of generics.
What About Market Risks?
- Medical Advancements: Development of alternative drugs with improved safety profiles may reduce reliance on digoxin.
- Safety Concerns: Narrow therapeutic window and risk of toxicity necessitate careful monitoring, possibly affecting prescribing habits.
- Regulatory Changes: Stricter labeling or restrictions could impede formulary inclusion.
- Patent and Patent Expiry: While no patents currently cover digoxin, changes in patent law or new formulations could influence pricing strategies.
Final Observations
- The digoxin market is mature, with low incremental growth.
- Emerging markets offer moderate growth opportunities due to increasing cardiovascular disease prevalence.
- Competitive pressure from newer agents limits the scope for significant market expansion in developed regions.
- Cost reductions via generics sustain sales but constrain profit margins.
Key Takeaways
- Global sales of digoxin are projected to grow at approximately 2% annually through 2027.
- The primary demand stems from established use in atrial fibrillation and heart failure management.
- Generic availability dominates sales, intensifying price competition.
- Market expansion hinges on growth in emerging economies and adherence to clinical guidelines.
- Risks include safety concerns and future availability of alternative therapies.
Frequently Asked Questions
1. How does the emergence of newer atrial fibrillation drugs impact digoxin?
Newer drugs, such as beta-blockers, calcium channel blockers, and antiarrhythmic agents, have expanded treatment options and sometimes offer better safety profiles. This limits digoxin's usage to specific cases and adjunct therapy.
2. Are there approved combination therapies involving digoxin?
Limited; digoxin is primarily used as monotherapy or adjunct therapy. No significant combination drugs are currently marketed, but research into adjunct use persists.
3. What are the key regulatory hurdles affecting digoxin sales?
Regulatory agencies demand rigorous labeling, especially concerning toxicity risks. Changes in safety guidelines can restrict prescribing or promote alternative treatments.
4. What is the outlook for digoxin in developing markets?
These markets present growth opportunities due to increasing cardiovascular disease burden and cost-sensitive healthcare. However, market penetration remains challenged by infrastructure issues and competing generic drugs.
5. Could patent protections or new formulations significantly alter the market?
Since digoxin is off-patent, patent-driven market shifts are unlikely. Innovations in formulations, such as extended-release versions, may offer minor growth avenues but won't drastically change total sales.
Sources
[1] MarketWatch. (2022). Global Digoxin Market Size, Share & Trends.
[2] Statista. (2022). Cardiovascular Disease Prevalence Worldwide.
[3] American College of Cardiology. (2022). Guideline Recommendations for Atrial Fibrillation Management.
[4] World Health Organization. (2021). Cardiovascular Diseases Fact Sheet.
[5] IMS Health. (2022). Global Generic Pharmaceutical Sales Data.