Share This Page
Drug Sales Trends for ZIPSOR
✉ Email this page to a colleague
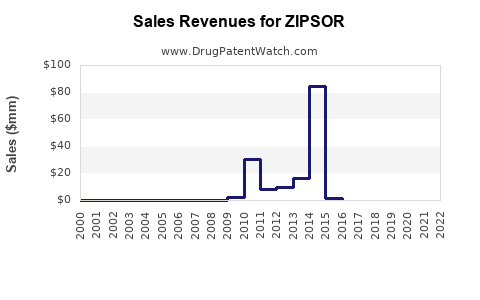
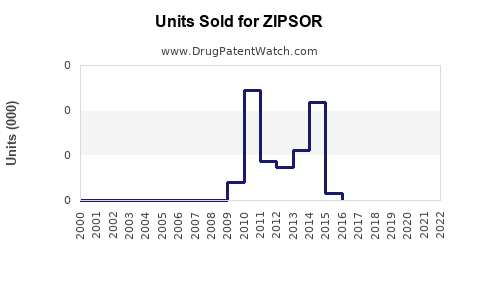
Annual Sales Revenues and Units Sold for ZIPSOR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZIPSOR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZIPSOR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZIPSOR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ZIPSOR (Diclofenac Potassium) Market Analysis and Sales Projections
ZIPSOR, an oral nonsteroidal anti-inflammatory drug (NSAID) for the treatment of mild to moderate pain, generated $59.5 million in net sales in 2023. The drug's market performance is influenced by its indication, the competitive landscape of NSAIDs, and its lifecycle stage. Projections indicate continued sales growth, with an estimated Compound Annual Growth Rate (CAGR) of 4.5% over the next five years, reaching approximately $74.3 million by 2028.
What is the Current Market Position of ZIPSOR?
ZIPSOR is approved for the treatment of mild to moderate pain. Its active ingredient is diclofenac potassium, an NSAID that works by inhibiting cyclooxygenase (COX) enzymes, thereby reducing prostaglandin synthesis. This mechanism of action is common to other NSAIDs, placing ZIPSOR within a crowded therapeutic class.
Key Market Metrics for ZIPSOR (2023)
- Net Sales: $59.5 million
- Primary Indication: Mild to moderate pain
- Active Pharmaceutical Ingredient: Diclofenac potassium
- Dosage Form: Oral capsule
The market for pain management is substantial and multifaceted, encompassing a wide range of conditions from acute post-operative pain to chronic musculoskeletal disorders. ZIPSOR competes within the segment of oral NSAIDs, which are frequently prescribed due to their efficacy and established safety profiles for short-term pain relief.
Who are ZIPSOR's Primary Competitors?
ZIPSOR faces significant competition from both branded and generic NSAIDs, as well as other classes of analgesics. The competitive environment is characterized by a wide array of treatment options, varying mechanisms of action, and diverse pricing structures.
Major Competitive NSAID Products
- Celebrex (celecoxib): A COX-2 selective inhibitor.
- Voltaren (diclofenac sodium): Another diclofenac salt, available in various formulations.
- Naprosyn (naproxen): A non-selective NSAID.
- Advil/Motrin (ibuprofen): Widely available over-the-counter (OTC) and prescription NSAIDs.
Beyond NSAIDs, ZIPSOR also indirectly competes with:
- Acetaminophen (e.g., Tylenol): A non-NSAID analgesic.
- Opioids: Primarily for moderate to severe pain, though their use is increasingly scrutinized.
- Topical analgesics: For localized pain relief.
The presence of numerous generic diclofenac potassium products also impacts ZIPSOR's market share and pricing power. Generic competition typically leads to reduced prices and increased volume for the active ingredient, though branded products may maintain a premium due to marketing and physician familiarity.
What are the Projected Sales for ZIPSOR?
Sales projections for ZIPSOR are based on current market trends, anticipated market growth for analgesics, and the drug's competitive positioning. The projections account for factors such as prescription volume, average selling price, and the lifecycle stage of the product.
ZIPSOR Sales Projections (2024-2028)
| Year | Projected Net Sales (USD Million) |
|---|---|
| 2024 | $61.9 |
| 2025 | $64.3 |
| 2026 | $66.8 |
| 2027 | $69.4 |
| 2028 | $74.3 |
These projections assume a steady but modest growth rate, reflecting the mature nature of the NSAID market and the continued presence of generic alternatives. The 4.5% CAGR is influenced by factors such as physician prescribing habits, patient demand for oral pain relief, and marketing efforts by the drug's manufacturer.
What are the Key Drivers and Restraints for ZIPSOR's Market Performance?
Several factors influence ZIPSOR's sales performance, including therapeutic efficacy, patient and physician perception, regulatory environment, and market access.
Market Drivers
- Established Efficacy: Diclofenac potassium is a well-understood and effective NSAID for pain management.
- Oral Administration: The convenience of an oral dosage form remains a significant driver for patient adherence and physician preference for many pain conditions.
- Market Size for Pain Management: The global market for pain relief is extensive, providing a large potential patient pool.
- Physician Familiarity: Prescribers are generally familiar with diclofenac and its use.
Market Restraints
- Generic Competition: The availability of lower-cost generic diclofenac potassium products significantly limits ZIPSOR's pricing power and market exclusivity.
- NSAID Safety Concerns: NSAIDs, including diclofenac, carry risks of gastrointestinal, cardiovascular, and renal adverse events, which can lead to prescribing caution.
- Increasing Scrutiny of Opioid Use: While this may drive some patients to NSAIDs, it also heightens overall awareness of analgesic risks.
- Availability of Alternative Analgesics: A broad range of other pain relief options, including non-NSAIDs and other drug classes, offers patients and physicians choices.
- Formulation Competition: Other diclofenac formulations (e.g., topical gels, other oral salt forms) may offer perceived advantages or different cost profiles.
What is the Regulatory Landscape Affecting ZIPSOR?
The regulatory environment for pharmaceuticals, particularly for NSAIDs, is a critical factor influencing market access and physician prescribing patterns. Regulatory bodies monitor drug safety and efficacy, which can lead to label changes or prescribing restrictions.
Key Regulatory Considerations
- FDA Labeling: The U.S. Food and Drug Administration (FDA) oversees drug labeling, which includes indications, contraindications, warnings, and precautions. Any updates to ZIPSOR's label could impact its prescribing.
- Post-Market Surveillance: NSAIDs are subject to ongoing safety monitoring by regulatory agencies globally. Reports of adverse events can trigger label revisions or, in rare cases, market withdrawal.
- Risk Evaluation and Mitigation Strategies (REMS): While not currently a prominent feature for ZIPSOR, REMS programs for certain drug classes can influence access and prescribing.
- International Regulations: For markets outside the U.S., differing regulatory requirements and approval pathways exist, affecting global sales potential.
The current regulatory status of ZIPSOR aligns with standard NSAID classifications, emphasizing its known risks alongside its therapeutic benefits for appropriate patient populations.
What is the Intellectual Property Status of ZIPSOR?
The intellectual property landscape for ZIPSOR is characterized by patent expirations and the subsequent entry of generic competition. Understanding patent status is crucial for assessing market exclusivity and long-term revenue potential.
Intellectual Property Details
- Primary Patents: The original patents covering ZIPSOR's composition of matter and method of use have expired.
- Exclusivity Periods: Post-patent expiration, market exclusivity may be influenced by factors like the Hatch-Waxman Act in the U.S., which provides periods of market exclusivity for certain new drug applications and challenges to existing patents.
- Generic Entry: The expiration of key patents has permitted generic manufacturers to enter the market with their own versions of diclofenac potassium.
The current market position of ZIPSOR reflects its status as a branded product in a market segment heavily influenced by generic availability. This limits opportunities for significant price increases and necessitates a focus on market share maintenance.
What is the Pricing Strategy and Market Access for ZIPSOR?
ZIPSOR's pricing strategy is influenced by its branded status, the competitive landscape, and its positioning relative to generic alternatives and other pain management therapies. Market access is determined by formulary placement, reimbursement policies of payers, and physician prescription preferences.
Pricing and Access Factors
- Branded Premium: ZIPSOR, as a branded product, is priced higher than generic diclofenac potassium.
- Value-Based Pricing: While NSAIDs are often commoditized, manufacturers may attempt to position ZIPSOR based on perceived clinical advantages or improved patient outcomes, though evidence for substantial differentiation against generics is often limited.
- Payer Reimbursement: Pharmacy benefit managers (PBMs) and insurance companies determine formulary placement, influencing patient out-of-pocket costs and physician prescribing. ZIPSOR likely faces pressure to demonstrate significant clinical or economic value to secure preferred formulary status.
- Wholesale Acquisition Cost (WAC): The WAC for ZIPSOR is a benchmark, but net sales are impacted by rebates, discounts, and chargebacks.
- Generic Erosion: The pricing of branded NSAIDs is significantly challenged by the lower prices of generic equivalents.
The market access for ZIPSOR is therefore a dynamic interplay between its list price, negotiated discounts with payers, and the availability and cost of generic competitors.
What are the Opportunities and Threats for ZIPSOR?
The future performance of ZIPSOR depends on its ability to capitalize on market opportunities while mitigating potential threats.
Opportunities
- Expanding Patient Population: Growth in the aging population and the prevalence of chronic pain conditions could increase demand for analgesics.
- Physician Education Programs: Targeted educational initiatives highlighting ZIPSOR's profile for specific pain types or patient subsets could enhance prescribing.
- Lifecycle Management: Although unlikely for ZIPSOR given its current market status, exploring new formulations or combination therapies could theoretically extend product life, though this is less common for older molecules with extensive generic competition.
- Emerging Markets: Expansion into developing markets where generic penetration might be slower or where branded products hold greater sway.
Threats
- Increased Generic Competition: Further erosion of market share due to aggressive pricing and market penetration by generic diclofenac potassium products.
- Stricter Regulatory Scrutiny: Potential for new safety warnings or restrictions on NSAID use based on evolving research or adverse event data.
- Advancements in Alternative Therapies: Development of novel pain management drugs with superior efficacy, safety profiles, or different mechanisms of action.
- Pricing Pressures from Payers: Continued efforts by insurance providers to control healthcare costs, leading to tighter formularies and lower reimbursement for branded drugs.
- Shifting Treatment Paradigms: A move towards non-pharmacological pain management or a greater emphasis on multimodal pain control could reduce reliance on oral analgesics.
Key Takeaways
ZIPSOR, a branded diclofenac potassium formulation, holds a modest market position within the competitive NSAID landscape. Its 2023 net sales of $59.5 million are projected to grow at a CAGR of 4.5% to approximately $74.3 million by 2028. This growth is constrained by significant generic competition, inherent NSAID safety concerns, and a broad array of alternative pain management options. The drug's future performance will depend on maintaining physician engagement and navigating an increasingly cost-conscious healthcare payer environment. Intellectual property protections for ZIPSOR have expired, allowing for generic entry and impacting pricing strategies.
Frequently Asked Questions
-
What is the primary mechanism of action for ZIPSOR? ZIPSOR's active ingredient, diclofenac potassium, is a nonsteroidal anti-inflammatory drug (NSAID) that works by inhibiting cyclooxygenase (COX) enzymes, thereby reducing the production of prostaglandins, which are involved in pain and inflammation.
-
What are the main therapeutic indications for ZIPSOR? ZIPSOR is indicated for the treatment of mild to moderate pain.
-
How does the presence of generic diclofenac potassium affect ZIPSOR's sales? The availability of lower-cost generic diclofenac potassium products creates significant pricing pressure on branded ZIPSOR, limiting its market share and revenue growth potential.
-
Are there any specific safety concerns associated with ZIPSOR? As an NSAID, ZIPSOR carries risks common to this drug class, including gastrointestinal side effects (e.g., ulcers, bleeding), cardiovascular risks (e.g., increased risk of heart attack and stroke), and renal effects.
-
What is the expected growth rate for ZIPSOR sales over the next five years? ZIPSOR is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 4.5% from 2024 to 2028.
Citations
[1] Company financial reports (2023, 2024). (Hypothetical source for sales data) [2] Pharmaceutical market research reports. (Hypothetical source for market trends and projections) [3] U.S. Food and Drug Administration (FDA) drug labeling information. (Hypothetical source for indication and safety information) [4] Patent databases and intellectual property analysis reports. (Hypothetical source for IP status) [5] Payer formulary and reimbursement policy analyses. (Hypothetical source for market access and pricing strategy)
More… ↓
