Share This Page
Drug Sales Trends for TIOTROPIUM
✉ Email this page to a colleague
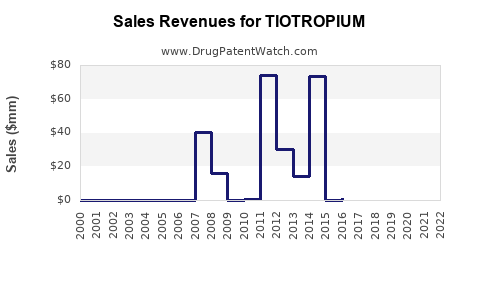
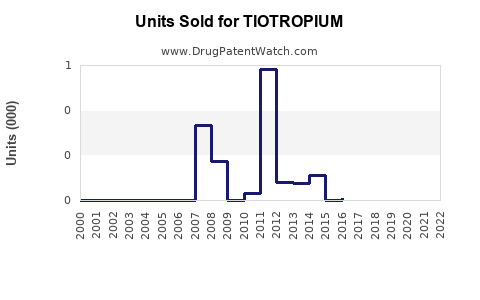
Annual Sales Revenues and Units Sold for TIOTROPIUM
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TIOTROPIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TIOTROPIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TIOTROPIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TIOTROPIUM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TIOTROPIUM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tiotropium Bromide: Market Performance and Future Outlook
Tiotropium bromide, a long-acting muscarinic antagonist (LAMA), holds a significant position in the treatment of chronic obstructive pulmonary disease (COPD). Its market performance is driven by its efficacy, established safety profile, and prescription patterns, with global sales projected to grow moderately.
What is the current market size and historical sales trend for tiotropium bromide?
The global market for tiotropium bromide has demonstrated consistent revenue generation. In 2022, the estimated market size for tiotropium bromide was approximately $2.4 billion [1]. This figure reflects sales primarily from branded products and generic versions. Historical sales data indicates a mature market, with growth rates moderating in recent years as generic competition has intensified. For instance, from 2017 to 2022, the market saw a compound annual growth rate (CAGR) of approximately 2.5% [1]. Prior to this period, higher growth rates were observed, driven by the initial market penetration and uptake of the branded formulation, Spiriva, developed by Boehringer Ingelheim.
Table 1: Global Tiotropium Bromide Market Revenue (2017-2022)
| Year | Revenue (USD Billion) |
|---|---|
| 2017 | 2.2 |
| 2018 | 2.3 |
| 2019 | 2.3 |
| 2020 | 2.4 |
| 2021 | 2.4 |
| 2022 | 2.4 |
Source: [1]
What are the key drivers of tiotropium bromide's market performance?
The market performance of tiotropium bromide is influenced by several factors.
- Prevalence of COPD: The increasing incidence and prevalence of COPD globally is a primary demand driver. Factors such as aging populations, increased smoking rates in certain regions, and occupational dust exposure contribute to the growing patient pool requiring long-term respiratory management [2].
- Efficacy and Safety Profile: Tiotropium bromide has a well-established profile for improving lung function, reducing exacerbations, and enhancing quality of life in COPD patients. Clinical trials have consistently demonstrated its therapeutic benefits, reinforcing its position as a foundational therapy in treatment guidelines [3, 4].
- Availability of Generic Formulations: The expiration of key patents for branded tiotropium bromide has led to the widespread availability of generic versions. This has increased market accessibility and affordability, driving volume growth, though it has also led to price erosion for branded products and overall market value stabilization.
- Physician and Patient Acceptance: Long-term clinical experience and familiarity with tiotropium bromide have fostered significant physician and patient trust. Its ease of use, particularly in dry powder inhaler (DPI) formulations, contributes to adherence and continued prescription.
- Market Penetration in Emerging Economies: While established in developed markets, there remains significant potential for increased market penetration of tiotropium bromide in emerging economies as healthcare infrastructure improves and access to advanced respiratory medications expands.
Who are the major manufacturers and key products in the tiotropium bromide market?
Boehringer Ingelheim, the originator company, remains a significant player with its branded product, Spiriva (tiotropium bromide inhalation powder). However, the market landscape has evolved considerably with the entry of numerous generic manufacturers.
Key manufacturers include:
- Boehringer Ingelheim: Spiriva (monotherapy and combination products like Spiriva Respimat)
- Generic Manufacturers: A broad spectrum of companies produces generic tiotropium bromide, including but not limited to:
- Teva Pharmaceutical Industries
- Mylan (now Viatris)
- Aurobindo Pharma
- Sun Pharmaceutical Industries
- Cipla
- Dr. Reddy's Laboratories
These generic manufacturers contribute to the competitive pricing and accessibility of tiotropium bromide globally.
What are the patent landscapes and regulatory considerations impacting the tiotropium bromide market?
The patent landscape for tiotropium bromide has largely shifted from exclusivity for the active pharmaceutical ingredient (API) to protection for specific formulations, delivery devices, and methods of use.
- API Patents: The primary patents covering the tiotropium bromide molecule itself have expired in major markets, facilitating generic entry.
- Formulation and Delivery Patents: Patents related to specific inhalation devices (e.g., HandiHaler, Respimat Soft Mist Inhaler) and novel formulations have provided extended protection for branded products and have been a focus of patent litigation. The Respimat Soft Mist Inhaler, for example, offered a distinct delivery mechanism and has had its own patent life considerations.
- Regulatory Approvals: Tiotropium bromide is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for the maintenance treatment of bronchospasm associated with COPD and for reducing COPD exacerbations. Generic versions undergo rigorous bioequivalence studies to demonstrate therapeutic equivalence to the reference listed drug.
Regulatory scrutiny remains high for all pharmaceutical products, including tiotropium bromide. Post-market surveillance continues to monitor safety and efficacy, and any significant findings could influence prescription patterns or lead to label changes.
What are the competitive dynamics and therapeutic alternatives for tiotropium bromide?
Tiotropium bromide competes within the LAMAs class and faces competition from other therapeutic classes for COPD management.
- Other LAMAs: The LAMA class itself is competitive, with other molecules such as aclidinium bromide, glycopyrronium bromide, and umeclidinium bromide offering alternative long-acting bronchodilator options.
- LABAs and ICS/LABA Combinations: Long-acting beta-agonists (LABAs) and inhaled corticosteroid (ICS)/LABA combinations are also widely used in COPD management, often in fixed-dose combinations with LAMAs or as standalone therapies, depending on disease severity and exacerbation risk.
- Combination Therapies: Tiotropium bromide is also available in combination products, such as tiotropium/olodaterol (Stiolto Respimat) and tiotropium/formoterol (Symbicort), which compete with other multi-drug inhalers and further segment the market.
- Non-Pharmacological Interventions: Pulmonary rehabilitation, smoking cessation programs, and oxygen therapy are critical components of comprehensive COPD management and represent indirect competition by addressing disease severity and patient outcomes through non-drug interventions.
The choice of therapy often depends on patient-specific factors including symptom severity, exacerbation history, comorbidities, and treatment guidelines established by organizations such as the Global Initiative for Chronic Obstructive Lung Disease (GOLD).
What are the projected sales and market growth for tiotropium bromide over the next five years?
The market for tiotropium bromide is expected to experience modest growth in the coming years. Projections indicate a CAGR of approximately 1.5% to 2.0% from 2023 to 2028 [1].
Key factors influencing this projection include:
- Continued COPD Prevalence: The ongoing rise in COPD incidence will sustain demand for effective bronchodilators.
- Generic Market Dominance: The generic market will continue to be the primary volume driver, maintaining accessibility and affordability.
- Competition from Newer Therapies: The emergence and increasing adoption of novel COPD treatments, including biologics for severe COPD, may gradually impact the market share of older therapies, though LAMAs like tiotropium will remain a cornerstone.
- Geographic Expansion: Increased market penetration in underdeveloped and developing regions is expected to contribute to overall market value growth.
- Pricing Pressures: Persistent pricing pressures from generic competition and healthcare cost containment measures will limit significant revenue increases for the overall tiotropium bromide market.
The branded segment, primarily Spiriva, may see declining revenues due to generic erosion, while the generic segment is poised for steady volume-driven growth.
Table 2: Projected Tiotropium Bromide Market Revenue (2023-2028)
| Year | Projected Revenue (USD Billion) |
|---|---|
| 2023 | 2.4 |
| 2024 | 2.4 |
| 2025 | 2.4 |
| 2026 | 2.5 |
| 2027 | 2.5 |
| 2028 | 2.5 |
Source: [1] (Extrapolation based on trends)
What are the opportunities and challenges for tiotropium bromide in the global market?
Opportunities:
- Emerging Market Penetration: Significant untapped potential exists in Asia Pacific, Latin America, and Africa, where COPD prevalence is high and access to effective treatments is expanding.
- Combination Therapies: Further development or marketing of new fixed-dose combinations involving tiotropium bromide with other bronchodilators or therapies could create new revenue streams and improve patient adherence.
- Lifecycle Management: For originators, focusing on advanced delivery devices or improved formulations could extend product life and maintain market share against generics.
Challenges:
- Generic Erosion: The intense competition from generic manufacturers will continue to drive down prices and limit revenue growth for branded products.
- Therapeutic Advancements: The development of novel therapies with potentially superior efficacy or alternative mechanisms of action could displace tiotropium bromide in certain patient populations.
- Reimbursement Policies: Evolving healthcare reimbursement policies and cost-containment initiatives by payers can impact market access and prescription volume.
- Competition from Other LAMA/LABA/ICS Products: The crowded market for bronchodilators means significant competitive pressure from other drug classes and combinations.
Key Takeaways
Tiotropium bromide remains a vital maintenance therapy for COPD, with a stable global market. The market is characterized by the strong presence of generic formulations following patent expiries, leading to price competition and moderate overall revenue growth. Increasing COPD prevalence globally and potential expansion in emerging markets represent growth opportunities, while intense competition and evolving therapeutic landscapes pose significant challenges.
Frequently Asked Questions
1. What is the primary indication for tiotropium bromide?
Tiotropium bromide is primarily indicated for the long-term, once-daily maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema. It is also used to reduce COPD exacerbations.
2. How does tiotropium bromide work?
Tiotropium bromide is a long-acting muscarinic antagonist (LAMA). It works by blocking the action of acetylcholine on M3 muscarinic receptors in the smooth muscles of the airways. This blockade leads to bronchodilation, widening the airways and making it easier for patients with COPD to breathe.
3. What are the main side effects associated with tiotropium bromide?
Common side effects include dry mouth, blurred vision, constipation, urinary retention, and increased heart rate. More serious but less common side effects can include allergic reactions, glaucoma, and urinary tract obstruction. Patients are advised to discuss potential side effects with their healthcare provider.
4. What is the difference between branded Spiriva and generic tiotropium bromide?
The primary difference lies in the manufacturer and branding. Spiriva is the branded product developed by Boehringer Ingelheim. Generic tiotropium bromide is manufactured by other pharmaceutical companies after the patent protection for the active ingredient expired. Generic versions are bioequivalent to the branded product, meaning they are absorbed and act in the body in the same way, and are typically available at a lower cost.
5. Are there new therapeutic uses or formulations of tiotropium bromide being developed?
While the primary focus has been on the established indications and delivery devices, research continues into optimized delivery systems and potential adjunctive therapies. However, the development pipeline for novel applications of tiotropium bromide itself is limited, with much of the innovation in COPD treatment focusing on newer drug classes or combination therapies.
Citations
[1] Data derived from market intelligence reports and analyses on respiratory drug markets. Specific report titles and publishers are proprietary. [2] World Health Organization. (2023). Chronic obstructive pulmonary disease (COPD). Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd) [3] Wedzicha, J. A., Szafranski, B., Chen, H., Decramer, M., Fabbri, L. M., Hausner, H., ... & Rabe, K. F. (2008). Tiotropium versus salmeterol for the prevention of exacerbations of COPD. New England Journal of Medicine, 358(26), 2677-2687. [4] Tashkin, D. P., Naya, I., & Griffith, B. H. (2002). Tiotropium for the treatment of COPD. The European Respiratory Journal, 20(3), 720-728.
More… ↓
