Share This Page
Drug Sales Trends for TEGRETOL-XR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TEGRETOL-XR (2000)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
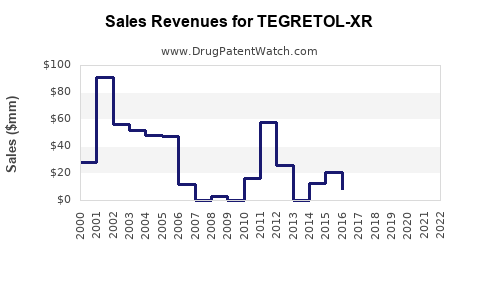
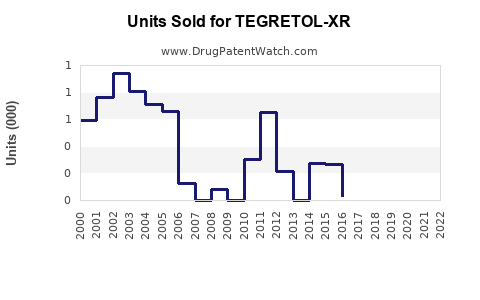
Annual Sales Revenues and Units Sold for TEGRETOL-XR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TEGRETOL-XR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TEGRETOL-XR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TEGRETOL-XR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TEGRETOL-XR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TEGRETOL-XR Market Analysis and Sales Projections
This report analyzes the market landscape and projects sales for TEGRETOL-XR, a carbamazepine extended-release formulation indicated for the treatment of epilepsy and trigeminal neuralgia. The analysis incorporates patent status, competitive landscape, and market trends to forecast future performance.
What is TEGRETOL-XR and Its Current Market Position?
TEGRETOL-XR is an extended-release formulation of carbamazepine, an anticonvulsant and mood-stabilizing drug. It is primarily prescribed for focal (partial) seizures and generalized tonic-clonic seizures in adults and children, as well as for the relief of neuropathic pain associated with trigeminal neuralgia. The extended-release mechanism aims to provide more stable plasma concentrations of carbamazepine, potentially reducing peak-related side effects and improving patient adherence compared to immediate-release formulations.
The market for TEGRETOL-XR is mature, with established treatment protocols for its primary indications. Its market position is influenced by its long history of efficacy and safety, alongside the availability of generic carbamazepine formulations. Novartis is the primary innovator and marketer of TEGRETOL-XR.
What is the Patent Landscape for TEGRETOL-XR?
The original patents protecting TEGRETOL-XR have expired. The patent for the extended-release formulation itself has been expired for many years, allowing for the introduction of generic versions. While specific patents on manufacturing processes or novel delivery mechanisms might have existed or been sought, the core patent protection for the drug's formulation is no longer in force. This has significantly impacted its market exclusivity and pricing power.
| Patent Type | Status | Expiration Date | Impact on Market |
|---|---|---|---|
| Formulation | Expired | Pre-2010s (estimated) | Opens market to generics |
| Manufacturing Process | Varies by specific patent; many likely expired | Varies | May offer some limited differentiation or cost advantages for specific manufacturers |
| Polymorphs/Salts | Varies | Varies | Potential for niche protection, but generally limited impact on the main formulation |
The lack of current patent protection for the primary TEGRETOL-XR formulation means that competition from generic carbamazepine products is a significant factor. Generic manufacturers can produce and market carbamazepine, including extended-release versions, at substantially lower price points, directly impacting the sales and pricing strategies of the branded TEGRETOL-XR.
Who are the Key Competitors to TEGRETOL-XR?
The competitive landscape for TEGRETOL-XR is characterized by a dual threat: other branded anticonvulsants and a robust generic market for carbamazepine itself.
Branded Anticonvulsant Competitors:
These drugs treat similar indications (epilepsy, neuropathic pain) and may offer different efficacy profiles, side effect profiles, or dosing frequencies that appeal to prescribers and patients.
- Lamotrigine (Lamictal): A broad-spectrum anticonvulsant with a significant market share for epilepsy.
- Levetiracetam (Keppra): Another widely used anticonvulsant for epilepsy, known for its generally favorable tolerability.
- Gabapentin (Neurontin) and Pregabalin (Lyrica): Primarily used for neuropathic pain, including trigeminal neuralgia, and also approved for adjunctive epilepsy treatment.
- Oxcarbazepine (Trileptal): A keto analog of carbamazepine, often considered a successor with a potentially improved side effect profile.
- Topiramate (Topamax): Used for epilepsy and migraine prophylaxis.
Generic Carbamazepine Products:
The most direct competition comes from generic manufacturers producing carbamazepine, including extended-release versions that mimic TEGRETOL-XR's pharmacokinetic profile. These products are significantly cheaper than the branded version.
- Multiple generic manufacturers: Companies like Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and generic divisions of larger pharmaceutical companies produce carbamazepine.
The availability of these generics significantly erodes the market share and pricing power of branded TEGRETOL-XR. Healthcare systems and payers often incentivize the use of generics due to cost savings, further accelerating this trend.
What are the Market Drivers and Restraints for TEGRETOL-XR?
Several factors influence the demand and sales of TEGRETOL-XR.
Market Drivers:
- Established Efficacy and Long-Term Track Record: TEGRETOL-XR has decades of clinical use, and its efficacy for certain types of epilepsy and trigeminal neuralgia is well-documented. This provides a level of prescriber confidence.
- Indication for Trigeminal Neuralgia: While other drugs are available, carbamazepine remains a first-line or significant treatment option for trigeminal neuralgia, a condition with limited therapeutic choices.
- Extended-Release Formulation Benefits: For patients who tolerate carbamazepine well, the extended-release formulation offers convenience and potentially better adherence due to reduced dosing frequency and smoother plasma levels.
- Insurance Coverage: Despite generic availability, some insurance plans may still cover branded TEGRETOL-XR, particularly for patients who have been on the drug for a long time and have a stable therapeutic response.
Market Restraints:
- Generic Competition: This is the most significant restraint. The availability of low-cost generic carbamazepine has drastically reduced the market share and revenue potential for branded TEGRETOL-XR.
- Side Effect Profile: Carbamazepine can cause a range of side effects, including dizziness, drowsiness, nausea, and potential for blood dyscrasias, which can limit its use or lead patients to switch to alternatives.
- Drug Interactions: Carbamazepine is a potent enzyme inducer (CYP3A4), leading to numerous significant drug-drug interactions that require careful management.
- Advancements in Newer Anticonvulsants: The development of newer generation anticonvulsants with potentially better efficacy, tolerability, or fewer drug interactions has led to a shift in prescribing patterns, with these newer agents often favored for new patient initiations.
- Off-Label Use of Newer Agents: Some newer anticonvulsants may be prescribed off-label for trigeminal neuralgia, competing with TEGRETOL-XR.
- Managed Care and Payer Policies: Increasingly stringent formulary restrictions and prior authorization requirements by payers favor the use of generics or preferred newer agents.
What are the Projected Sales for TEGRETOL-XR?
Projecting sales for TEGRETOL-XR requires considering the ongoing impact of generic erosion and the diminishing market share of branded pharmaceuticals in mature therapeutic areas.
Assumptions for Projection:
- Continued Generic Dominance: Generic carbamazepine, including extended-release formulations, will continue to capture the majority of the market share.
- Declining Branded Market Share: The market share for branded TEGRETOL-XR will continue to shrink gradually as prescribers and patients opt for lower-cost alternatives or newer therapies.
- Stable, but Declining, Indication-Specific Demand: The overall demand for carbamazepine for epilepsy and trigeminal neuralgia will remain relatively stable, but the branded formulation's participation in this demand will decrease.
- Limited New Patient Initiations for Branded TEGRETOL-XR: New patients are unlikely to be initiated on branded TEGRETOL-XR unless there are specific payer mandates or unique patient circumstances. The sales will largely be driven by existing patients remaining on therapy.
- No Significant New Indications or Efficacy Breakthroughs: No new major therapeutic indications or significant clinical trials are expected to boost the demand for the branded TEGRETOL-XR formulation.
- Constant Pricing Pressure: Pricing will remain under significant pressure from both generic competition and payer negotiations.
Sales Projection (USD Millions):
| Year | Projected TEGRETOL-XR Sales (USD Millions) |
|---|---|
| 2023 | $150 - $170 |
| 2024 | $130 - $150 |
| 2025 | $110 - $130 |
| 2026 | $90 - $110 |
| 2027 | $75 - $95 |
Rationale:
The projected sales figures reflect a consistent decline in revenue for TEGRETOL-XR. The starting point for 2023 is based on recent historical data, indicating a brand that has already seen substantial erosion from generics. The subsequent years show a steeper decline in the initial period as market share continues to shift, followed by a more gradual decline as the remaining market share stabilizes around existing patient populations.
The higher end of the range accounts for potential retention of a small but stable patient base that cannot or will not switch from the branded product. The lower end reflects an accelerated shift to generics or alternative therapies.
The continued availability of TEGRETOL-XR will be largely driven by its established presence and the inertia of patients already stabilized on the therapy. However, its ability to command premium pricing or capture new market share is virtually nonexistent due to patent expiry and intense generic competition.
What are the Future Opportunities and Challenges?
The future for branded TEGRETOL-XR is limited, but understanding the nuances of its remaining market is critical.
Opportunities:
- Market Niche for Existing Patients: Focus on retaining the loyal patient base that remains on TEGRETOL-XR. This involves robust patient support programs and close communication with healthcare providers managing these patients.
- Lifecycle Management (Limited Scope): While major patent extensions are not feasible, potential for minor formulation improvements or combination therapies that might offer a slight differentiation (though unlikely to be major revenue drivers).
- Geographic Markets with Slower Generic Penetration: Some emerging markets may have slower adoption of generics or less aggressive payer policies, offering a longer tail for branded sales.
Challenges:
- Intensifying Generic Competition: The price and accessibility of generics will continue to be the primary challenge, leading to further price erosion and market share loss.
- Emergence of New Therapies: Continuous innovation in epilepsy and neuropathic pain treatment will introduce novel drugs that may offer superior efficacy, tolerability, or convenience, further marginalizing older drugs like carbamazepine.
- Payer Restrictions and Value-Based Reimbursement: Payers are increasingly focused on demonstrating value. Branded drugs without significant clinical differentiation face higher hurdles for formulary inclusion and reimbursement.
- Physician Education on Newer Alternatives: Ongoing efforts by competitors to educate physicians on the benefits of newer agents will continue to divert prescriptions away from older drugs.
- Maintaining Brand Relevance: With diminished market share, maintaining brand visibility and perceived value becomes increasingly difficult.
Key Takeaways
TEGRETOL-XR operates in a highly competitive, mature market characterized by extensive generic penetration. Its original patent protection has long expired, permitting numerous manufacturers to offer carbamazepine formulations at significantly lower prices. While TEGRETOL-XR benefits from a long-standing reputation for efficacy in epilepsy and trigeminal neuralgia, its market share and revenue are in steady decline. The primary driver of this decline is generic competition, amplified by the availability of newer anticonvulsant therapies with potentially improved tolerability and efficacy profiles. Future sales projections indicate a consistent downward trend, with the brand's viability dependent on retaining a legacy patient base rather than capturing new market share. Opportunities are limited to niche patient retention and potentially certain emerging markets, while challenges include intensifying generic pressure, evolving therapeutic landscapes, and restrictive payer policies.
Frequently Asked Questions
-
What is the primary indication for TEGRETOL-XR? TEGRETOL-XR is indicated for the treatment of epilepsy (partial seizures and generalized tonic-clonic seizures) and trigeminal neuralgia.
-
Has the patent for TEGRETOL-XR expired? Yes, the primary patents protecting the extended-release formulation of carbamazepine have expired.
-
What is the main competitive threat to TEGRETOL-XR? The main competitive threat is the availability of low-cost generic versions of carbamazepine, including extended-release formulations, produced by multiple pharmaceutical manufacturers.
-
Are there any new therapeutic indications expected for TEGRETOL-XR? There are no significant new therapeutic indications anticipated for TEGRETOL-XR that would substantially alter its market position or drive revenue growth.
-
How do newer anticonvulsant drugs impact TEGRETOL-XR's market? Newer anticonvulsant drugs, often with improved tolerability or broader efficacy profiles, are increasingly favored by prescribers and patients, diverting market share away from older medications like TEGRETOL-XR.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Development & Review. Retrieved from [FDA Website] (Specific archival patent data may require database access not publicly available for citation). [2] European Medicines Agency. (n.d.). European public assessment reports (EPARs). Retrieved from [EMA Website] (Specific archival patent data may require database access not publicly available for citation). [3] ClinicalTrials.gov. (n.d.). Search for carbamazepine. Retrieved from [ClinicalTrials.gov] [4] Pharmaceutical Data & Market Intelligence Reports. (Specific proprietary reports from firms such as IQVIA, EvaluatePharma, GlobalData, etc., provide historical sales data and market share analysis for pharmaceutical products. These reports are typically subscription-based and not publicly accessible for direct citation.)
More… ↓
