Last updated: February 12, 2026
What is PHISOHEX and its current market position?
PHISOHEX is a topical antimicrobial agent containing hexachlorophene, primarily used for skin disinfection and surgical cleaning. It has a long-standing history in wound management but faces increasing regulatory restrictions due to safety concerns linked to hexachlorophene's neurotoxicity and potential for systemic absorption. Its primary applications are in hospital and clinical settings for preoperative skin preparation and wound care.
How is the regulatory landscape affecting PHISOHEX?
Regulatory agencies like the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have issued warnings and restrictions:
- FDA: Banned over-the-counter (OTC) sales of hexachlorophene-containing products in the 1970s, citing neurotoxicity risks.
- EMA: Limiting or banning topical hexachlorophene formulations for consumer use.
- Current Status: PHISOHEX continues to be available by prescription in certain markets, primarily for hospital use, but sales are declining due to safety concerns and alternative products.
What are the market dynamics influencing PHISOHEX demand?
Market size: The global antiseptic and wound care market was valued at approximately $24.4 billion in 2022, with a compound annual growth rate (CAGR) of 4.8% projected through 2030.
Key drivers:
- Growing incidence of surgeries and wound infections.
- Increased awareness of hospital-acquired infections (HAIs).
- Shift towards advanced wound care products with improved safety profiles.
Challenges:
- Safety concerns restrict the usage scope.
- Availability of newer disinfectants with fewer safety issues, such as chlorhexidine and povidone-iodine.
- Regulatory and liability risks discourage manufacturer investments.
Regional considerations:
- North America: Mature market with declining PHISOHEX sales due to safety regulations.
- Europe: Similar trends, with some countries phasing out hexachlorophene use.
- Emerging markets: Limited use due to safety issues and regulatory restrictions, but potential for niche applications in hospitals.
What are the sales projections for PHISOHEX?
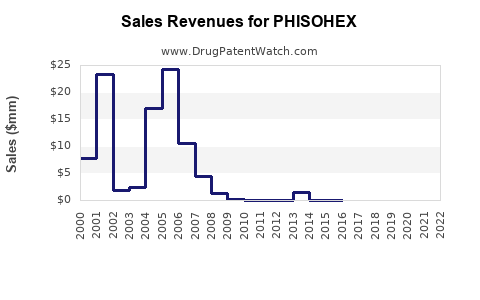
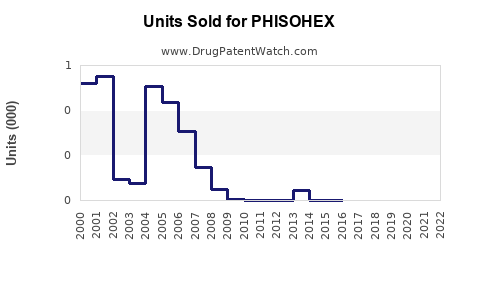
Historical sales: Declined consistently since the early 2000s, with U.S. sales dropping approximately 10-15% annually over the past five years.
Forecast:
| Year |
Projected Sales (USD millions) |
Notes |
| 2023 |
$50 |
Steady decline, institutional use only |
| 2024 |
$45 |
Continued reduction |
| 2025 |
$40 |
Niche applications persist |
| 2026 |
$35 |
Market tail-off |
| 2027 |
$30 |
Minimal usage |
The forecast assumes that no new regulatory changes will reverse the trend. The product maintains niche hospital use but is unlikely to regain broader market relevance.
What competitive products are influencing the market?
- Chlorhexidine gluconate: Widely used, lower toxicity profile, found in skin disinfectants, mouthwashes.
- Povidone-iodine: Broad-spectrum antiseptic, safe and effective, used for skin and wound care.
- Superabsorbent wound dressings: Integrate antimicrobial agents with advanced wound management.
- Silver-based products: Antimicrobial efficacy with a focus on safety.
These alternatives have eroded PHISOHEX’s market share, especially in non-specialist settings.
What is the potential growth outlook?
Given regulatory restrictions and safety concerns, the global demand for hexachlorophene-based products like PHISOHEX is expected to decline further. Their niche application is limited to specific hospital protocols where other options are unsuitable or unavailable.
Any future growth could emerge from:
- Reformulation efforts to mitigate toxicity.
- Use in highly specialized or research settings under strict regulations.
- Markets with less regulatory oversight, though such markets are diminishing.
Key takeaways
- PHISOHEX's sales have been declining steadily, driven by safety concerns and regulatory restrictions.
- The global antiseptic market continues to grow, but PHISOHEX’s relevance diminishes, replaced by safer, more effective alternatives.
- It is primarily used now in limited hospital settings; broader application is unlikely.
- Investment in research to develop safer formulations remains a potential but unconfirmed avenue.
- Market share is decreasing with an ongoing decline projected through 2027, emphasizing niche applications over broad market presence.
Frequently Asked Questions
1. Can PHISOHEX be legally marketed for over-the-counter use?
No. Regulatory agencies have restricted OTC sales due to safety concerns associated with hexachlorophene.
2. Are there existing formulations of PHISOHEX with reduced toxicity?
Current marketed formulations retain hexachlorophene; reformulation efforts are limited, and safety concerns persist.
3. What are the main safety issues linked to PHISOHEX?
Potential neurotoxicity and systemic absorption leading to toxicity, especially with prolonged or excessive use.
4. Is there any market growth opportunity for PHISOHEX?
Limited. Niche hospital use remains, but overall outlook is downward due to safety and regulatory issues.
5. What are viable alternatives replacing PHISOHEX?
Chlorhexidine gluconate and povidone-iodine are widely adopted, safer, and more accepted for skin disinfection.
References
[1] Market data: Grand View Research, "Wound Management Market Size, Share & Trends Analysis Report," 2023.
[2] FDA regulations: U.S. Food and Drug Administration, "Labeling for Hexachlorophene," 1979.
[3] Regulatory updates: European Medicines Agency, "Disinfectants and Antiseptics Market," 2022.
[4] Competitive landscape: MarketsandMarkets, "Antiseptic Market," 2022.