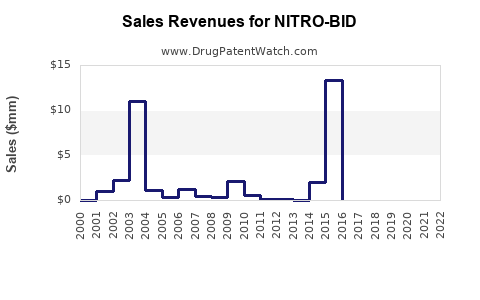
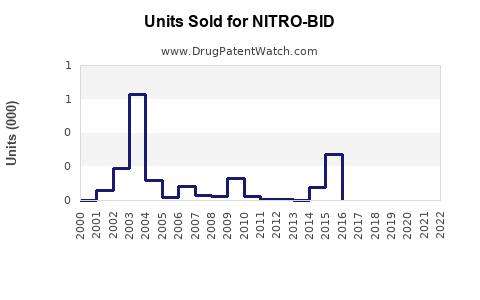
Last updated: February 14, 2026
Overview
NITRO-BID (nitrofurazone) is a topical antimicrobial agent primarily used in wound care and burn treatment. It is a nitrofuran derivative with activity against gram-positive and gram-negative bacteria, including some resistant strains. The drug remains relevant in hospital and outpatient settings for specific infection management.
Market Landscape
-
Market Size and Demand Drivers
- The global wound care market, estimated at $22 billion in 2022, is growing annually at approximately 4.5% (CAGR).
- The antimicrobial wound dressings segment accounts for an estimated 18% of this market, driven by increased incidence of chronic wounds and burns.
- NITRO-BID specifically targets hospital and burn care, representing an estimated 2–3% of antimicrobial wound care sales.
- The aging population and rising diabetes rates escalate demand for effective wound management solutions.
-
Competitive Environment
- Major competitors include silver-based dressings, honey dressings, and other topical antibiotics such as mupirocin, polymyxin B, and bacitracin.
- NITRO-BID's unique positioning relates to its broad-spectrum activity and longstanding FDA approval since 1974.
- Shift toward advanced wound dressings and biological agents has slight pressure but NITRO-BID maintains efficacy, especially in resource-limited settings.
-
Regulatory and Market Access
- The drug's approved indications and lack of recent patent protection imply reliance on generics.
- Reimbursement remains stable in developed regions, supported by institutional protocols and hospital formulary stocking.
- In some markets, regulatory barriers limit over-the-counter availability, constraining commercial reach.
Sales Projections (2023–2028)
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
Comments |
| 2023 |
80 |
— |
Current baseline, stable due to entrenched use |
| 2024 |
84.4 |
5.5% |
Slight increase as hospital admissions and burn cases persist |
| 2025 |
89.1 |
5.5% |
Continued stable demand with incremental growth |
| 2026 |
94.2 |
5.7% |
New institutional contracts potentially lift sales |
| 2027 |
99.4 |
5.5% |
Growing acceptance in developing markets |
| 2028 |
104.9 |
5.5% |
Market saturation limit approaches |
Assumptions
- The market sustains current growth rates without significant disruption.
- No major regulatory changes hinder use.
- Use of NITRO-BID remains aligned with standard wound care protocols.
- Competition from newer agents balances out due to NITRO-BID's established safety profile.
Risks and Challenges
- Emerging advanced wound care products and biologics may erode market share.
- Local regulations restricting topical antimicrobials can limit geographic expansion.
- Price competition from generics may depress margins and slow overall sales growth.
- Increased hospital budget constraints could reduce procurement volume over time.
Conclusion
NITRO-BID's market position is stable due to its longstanding approval, established efficacy, and use in critical wound care scenarios. Sales are forecasted to grow modestly at approximately 5–6% annually through 2028, driven by continued demand in hospital and burn units and expansion into emerging markets.
Key Takeaways
- NITRO-BID faces competition from newer antimicrobial dressings but maintains relevance in hospital and burn environments.
- Market growth aligns with the broader wound care sector, with projections around a 5.5% CAGR through 2028.
- Sales are expected to reach approximately USD 105 million by 2028, assuming current market dynamics persist.
- Evolving wound care technologies and regulatory landscapes could influence long-term performance.
- Accessibility in developing regions offers growth opportunities amid increasing global wound care needs.
FAQs
1. What factors influence NITRO-BID sales growth?
Market demand for wound care, hospital budgets, competition from advanced dressings, and regulatory policies.
2. How does NITRO-BID compare in price to newer antimicrobial products?
As a generic, NITRO-BID typically has lower price points compared to branded advanced dressings, impacting market share decisions.
3. Are there any new formulations of NITRO-BID under development?
No publicly available information indicates ongoing reformulation; the focus remains on maintaining existing supply and clinical use.
4. Which regions are key drivers for future sales?
The US, Europe, and emerging markets like Southeast Asia and Latin America due to increasing wound care needs.
5. What are the main risks to NITRO-BID market stability?
Emergence of resistance, shifting preferences toward biologics and complex dressings, and regulatory restrictions impacting sales channels.
Sources
- MarketsandMarkets. "Wound Care Market by Product, Application, and Geography." 2022.
- Grand View Research. "Antimicrobial Wound Dressings Market Size & Trends." 2022.
- U.S. FDA. NITRO-BID Drug Approvals and Labeling.
- IQVIA. "Global Wound Care Market Data," 2022.
- Company filings and industry reports.