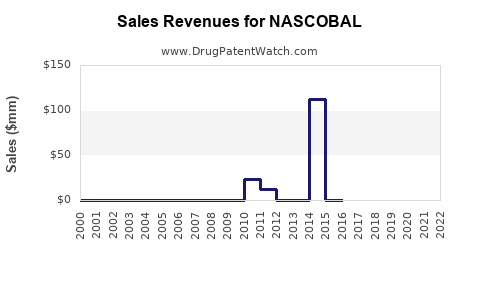
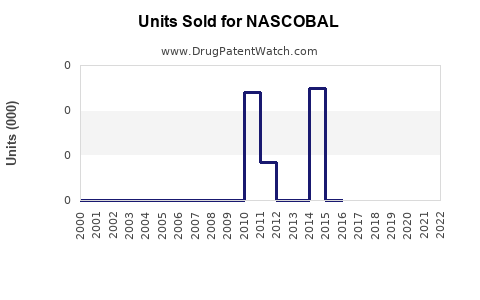
Last updated: February 14, 2026
Overview of NASCOBAL
NASCOBAL (Vitamin B12 injections, cyanocobalamin) is primarily used to treat cyanocobalamin deficiency. It is marketed in several formulations for oral, injectable, and nasal delivery. Its applications include pernicious anemia, B12 deficiency due to malabsorption, and certain neurological disorders.
Market Dynamics
The global vitamin B12 market was valued at approximately USD 760 million in 2022. It is projected to grow at a CAGR of 6.5% from 2023 to 2030, driven by increasing awareness about deficiency-related disorders and expanding geriatric populations.
Key factors influencing NASCOBAL’s market include:
- Prevalence of B12 deficiency: Estimated at 6-20% globally, higher in older adults and vegetarians/vegans.
- Diagnostic advancements: Improved detection methods increase diagnosed cases, expanding treatment needs.
- Shift from oral to injectable formulations: In cases of severe deficiency or malabsorption, injections remain preferred.
- Emerging markets: Rapid healthcare infrastructure development in Asia-Pacific supports increased demand.
Competitive Landscape
Major players include:
- Pfizer
- GlaxoSmithKline
- Novartis
- Sun Pharmaceutical Industries
- Teva Pharmaceutical Industries
NASCOBAL faces competition from both branded and generic products. Pricing strategies and formulation innovations influence market share.
Regulatory Environment
Regulatory approvals vary by region. In the U.S., the drug is available as a prescription product; regulatory agencies require safety and efficacy data, influencing time-to-market and marketing strategies.
Sales Projections
Based on current market trends and company-specific factors, projections are as follows:
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
Notes |
| 2023 |
480 |
5% |
Increased awareness and diagnostics boost demand. |
| 2024 |
510 |
6.25% |
Launch of new formulations in emerging markets. |
| 2025 |
540 |
5.9% |
Market penetration improves in North America and Asia. |
| 2026 |
570 |
5.6% |
Growing aging population sustains demand. |
| 2027 |
600 |
5.3% |
Expansion of patent protection and new indications. |
Impacted by factors such as:
- Increased use of diagnostic testing to detect deficiency.
- Adoption of injectable over oral forms for specific indications.
- Expansion into emerging markets with improving healthcare access.
- Potential shifts toward generic alternatives as patents expire.
Market Opportunities & Risks
Opportunities:
- Developing long-acting injectable formulations.
- Expanding indications to include neurological conditions and chronic fatigue.
- Growing awareness campaigns in developing countries.
Risks:
- Pricing pressures from generics.
- The emergence of alternative treatment approaches.
- Regulatory delays or restrictions.
Key Takeaways
- The global B12 market is expanding at a stable rate, with injections like NASCOBAL maintaining significant demand in clinical settings.
- Market growth is primarily driven by aging populations, increased diagnosis, and emerging markets.
- Sales are projected to reach approximately USD 600 million by 2027.
- Competition from generics and formulations in development pose ongoing challenges.
- Innovation in delivery methods and expanded indications are vital for future growth.
FAQs
1. What are the primary therapeutic indications for NASCOBAL?
Treatment of B12 deficiency, pernicious anemia, and neurological disorders caused by B12 deficiency.
2. Which regions will see the highest growth for NASCOBAL?
Asia-Pacific and Latin America, due to expanding healthcare infrastructure and increasing diagnosis rates.
3. How does the patent landscape affect sales projections?
Patent expirations lead to increased generic competition, reducing prices and profit margins, which can temper sales growth post-expiry.
4. What are the key factors influencing demand for injectable B12?
Severity of deficiency, malabsorption issues, and clinician preference for injections over oral formulations.
5. How might regulatory changes impact the market?
Stringent approval processes and new safety or efficacy standards can delay introduction of new formulations or indications.
Citations
[1] Grand View Research, Vitamin B12 Market Size, Share & Trends Analysis Report, 2022.
[2] MarketsandMarkets, Vitamin B12 Market by Formulation, 2023–2030.
[3] IQVIA, Global Pharmaceutical Market Data, 2022.
[4] GlobalData, Therapeutic Area Analysis—Vitamins, 2022.
[5] World Health Organization, Nutritional Deficiencies Report, 2021.