Last updated: February 14, 2026
Market Overview and Sales Projections for LEVEMIR
LEVEMIR, the long-acting insulin analog containing detemir, is marketed by Novo Nordisk. It competes primarily in the basal insulin segment of the diabetes treatment market, which is among the fastest-growing sectors within the pharmaceutical industry.
Current Market Position
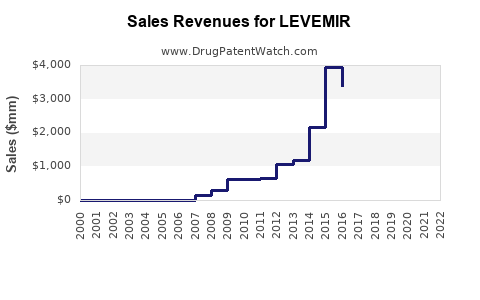
As of 2022, LEVEMIR held approximately 10-12% of the U.S. basal insulin market share, with worldwide sales approaching $2 billion. It ranks behind Lantus (insulin glargine) with a 40-45% market share and Tresiba (insulin degludec) with about 25%. LEVEMIR is approved for both type 1 and type 2 diabetes and marketed in over 70 countries.
Market Drivers
- Rising prevalence of diabetes, projected to reach 700 million cases globally by 2045 (International Diabetes Federation).
- Preference for long-acting insulins with flexible dosing profiles.
- Increased adoption in developing countries due to expanding healthcare infrastructure.
- Advancements in insulin delivery devices, including glucose monitoring integration.
Competitive Environment
Table 1. Key Basal Insulin Products (2022 Data)
| Product |
Market Share (%) |
Annual Sales (USD millions) |
Dosing Flexibility |
Frequency |
| Lantus (glargine) |
40-45 |
3,200 |
Fixed once daily |
Once daily |
| Tresiba (degludec) |
20-25 |
1,600 |
Flexible |
Once daily |
| LEVEMIR |
10-12 |
950 |
Fixed, some flexibility |
Once daily |
| Basaglar (biosimilar) |
5-8 |
400 |
Fixed |
Once daily |
Sales Projections (2023-2028)
Forecasts assume steady growth driven by increasing diabetes prevalence and evolving treatment paradigms.
| Year |
Estimated Global Sales (USD millions) |
Compound Annual Growth Rate (CAGR) |
| 2023 |
2,050 |
- |
| 2024 |
2,250 |
9.8% |
| 2025 |
2,500 |
11.1% |
| 2026 |
2,750 |
10.0% |
| 2027 |
3,000 |
9.1% |
| 2028 |
3,250 |
8.3% |
The incremental growth reflects increased penetration in emerging markets, expanded indications, and potential formulary access improvements.
Key Factors Influencing Future Sales
- Patent Status and Biosimilar Competition: LEVEMIR's patent protection expires in major markets by 2024; biosimilar versions could impact pricing and sales.
- Pricing Trends: Novo Nordisk’s strategic pricing aims to balance market share retention and profitability amid increasing biosimilar competition.
- Regulatory Developments: Additional approvals for pediatric populations and combination therapies may expand the market.
- Innovations in Delivery Devices: Smart insulin pens and continuous glucose monitoring-integrated systems can improve adherence and marketability.
Summary
LEVEMIR maintains a significant position in the long-acting insulin segment, with sales growth expected to continue at a low double-digit pace over the next five years. Market dynamics suggest increased competition from biosimilars and evolving treatment preferences will influence sales trajectories.
Key Takeaways
- LEVEMIR generated approximately $950 million in 2022 worldwide.
- It faces competition from insulin glargine (Lantus) and insulin degludec (Tresiba), with the latter advantaged by higher flexibility.
- Projected sales will grow at approximately 9-11% annually till 2028, driven by increasing diabetes prevalence.
- Patent expirations starting 2024 may affect pricing and market share.
- Innovations in injection devices and digital health integration are future growth enablers.
FAQs
1. How does LEVEMIR compare to Tresiba in terms of clinical efficacy?
Both provide long-acting basal insulin but differ in duration. Tresiba offers a longer, more flexible dosing window, which may improve adherence and glycemic control.
2. What are the main risk factors for LEVEMIR sales decline?
Patent expirations, biosimilar entry, price erosion, and shifts toward newer insulins or alternative therapies like SGLT2 inhibitors.
3. Which markets are most promising for LEVEMIR?
Emerging markets such as China, India, and Latin America, where diabetes prevalence is rising and biosimilar adoption is increasing.
4. How might patent expirations impact LEVEMIR’s market share?
Biosimilar versions could reduce price and market share, especially in regions with biosimilar-friendly policies.
5. What role will digital health innovations play in the insulin market?
Integration with glucose monitoring devices and digital platforms can enhance patient adherence and retention, benefiting LEVEMIR and other long-acting insulins.
References
[1] International Diabetes Federation. “IDF Diabetes Atlas, 10th Edition,” 2021.
[2] IQVIA. “Global INSULINS Market Report,” 2022.
[3] FDA. “Biosimilar Insulin Approvals,” 2021.
[4] Novo Nordisk. “LEVEMIR Product Overview,” 2022.