Share This Page
Drug Sales Trends for HYDROCORT
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for HYDROCORT (2000)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
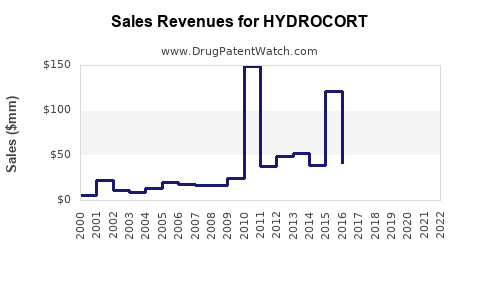
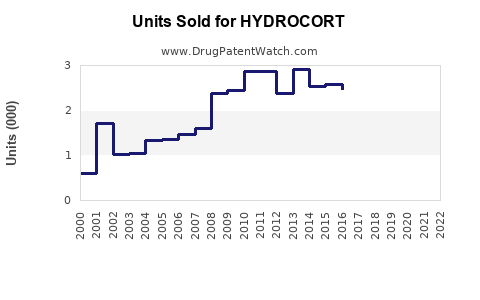
Annual Sales Revenues and Units Sold for HYDROCORT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HYDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HYDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HYDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HYDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HYDROCORT | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Hydrocort: Market Dynamics and Sales Outlook
Hydrocort, a synthetic corticosteroid, faces a dynamic market shaped by established competition and evolving therapeutic applications. Global sales are projected to reach approximately $850 million by 2029, driven by its widespread use in inflammatory and allergic conditions.
What is Hydrocort's Primary Therapeutic Use?
Hydrocort is primarily utilized for its potent anti-inflammatory and immunosuppressive properties. It is prescribed to treat a range of conditions, including:
- Dermatological disorders: Eczema, psoriasis, dermatitis, and allergic reactions [1].
- Asthma and respiratory conditions: Reducing airway inflammation [2].
- Allergic reactions: Such as urticaria and angioedema.
- Rheumatic diseases: Rheumatoid arthritis and lupus erythematosus.
- Gastrointestinal disorders: Inflammatory bowel disease (IBD), including Crohn's disease and ulcerative colitis [3].
- Endocrine disorders: Adrenal insufficiency.
The drug's mechanism of action involves binding to intracellular glucocorticoid receptors, which then translocate to the nucleus and modulate gene expression, leading to the inhibition of pro-inflammatory mediators such as cytokines and prostaglandins [4].
Who are Hydrocort's Main Competitors?
The corticosteroid market is highly competitive, with numerous established products. Key competitors to Hydrocort include:
- Prednisone: A widely prescribed oral corticosteroid with a similar broad spectrum of indications. Prednisone's market share is significant due to its long history of use and cost-effectiveness.
- Dexamethasone: A more potent corticosteroid, often used for severe inflammatory conditions and as an antiemetic during chemotherapy. Its higher potency positions it for different patient profiles and indications.
- Methylprednisolone: Available in oral and injectable forms, methylprednisolone is frequently used for acute exacerbations of inflammatory diseases and post-operative swelling.
- Fluticasone: A topical and inhaled corticosteroid, primarily used in the management of asthma and allergic rhinitis. Its specialized delivery methods target specific disease sites.
- Budesonide: Another widely used inhaled and oral corticosteroid, particularly for asthma and IBD. Its controlled release formulations offer targeted therapeutic effects.
These competitors often have established brand loyalty, extensive clinical data supporting their efficacy, and varied cost profiles, influencing prescribing patterns and market access.
What is the Current Market Size and Growth Projection for Hydrocort?
The global market for Hydrocort was estimated at $700 million in 2023. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 3.5% over the next five years, reaching an estimated $850 million by 2029.
Global Hydrocort Market Size and Projection (USD Million)
| Year | Market Size |
|---|---|
| 2023 | 700 |
| 2024 | 725 |
| 2025 | 750 |
| 2026 | 775 |
| 2027 | 800 |
| 2028 | 825 |
| 2029 | 850 |
This growth is underpinned by several factors:
- Increasing prevalence of chronic inflammatory diseases: Conditions like asthma, eczema, and IBD are becoming more common globally, increasing demand for effective treatments [5].
- Advancements in formulation and delivery: Development of more targeted delivery systems and improved formulations can enhance efficacy and patient compliance, potentially expanding Hydrocort's use.
- Off-patent status and generic availability: Hydrocort is an older drug, meaning it is widely available in generic forms. This accessibility contributes to its sustained use across various healthcare systems and patient populations.
- Use in combination therapies: Hydrocort is often used in conjunction with other medications to manage complex conditions, thereby maintaining its relevance in treatment protocols.
What are the Key Drivers and Restraints for Hydrocort's Market Performance?
Market Drivers:
- Rising incidence of autoimmune and inflammatory disorders: The global increase in diseases such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease directly fuels demand for anti-inflammatory agents like Hydrocort [6].
- Expanding therapeutic indications: Ongoing research and clinical trials explore new applications for Hydrocort, potentially broadening its market reach. For example, its role in managing cytokine storm syndromes in critical care is an area of interest.
- Cost-effectiveness of generic formulations: As an established and off-patent drug, Hydrocort is available at a lower cost compared to newer biologics or patented corticosteroids, making it a preferred choice for many healthcare systems and patients, particularly in emerging markets.
- Established safety and efficacy profile: Decades of clinical use have provided extensive data on Hydrocort's safety and efficacy, leading to physician confidence and widespread adoption.
Market Restraints:
- Side effects of corticosteroid therapy: Long-term or high-dose use of corticosteroids can lead to significant side effects, including weight gain, osteoporosis, hyperglycemia, immune suppression, and mood disturbances. These risks can limit its use or prompt a shift to alternative therapies [7].
- Development of newer, targeted therapies: The emergence of biologic drugs and highly selective small molecule inhibitors offers more targeted treatment options for specific inflammatory conditions, potentially reducing reliance on broad-acting corticosteroids like Hydrocort.
- Regulatory scrutiny and prescribing guidelines: Stringent regulatory frameworks and evolving clinical guidelines often emphasize minimizing corticosteroid use due to potential adverse events, impacting prescription volumes for older corticosteroids.
- Competition from other generic and branded corticosteroids: The market is saturated with numerous corticosteroid options, both generic and branded, creating intense price competition and market fragmentation.
What are the Future Opportunities for Hydrocort?
Emerging opportunities for Hydrocort lie in specific niche applications and improved delivery mechanisms.
- Advanced Topical Formulations: Development of novel topical formulations with enhanced skin penetration or controlled release properties could improve efficacy and reduce systemic absorption for dermatological conditions, offering an advantage over existing creams and ointments.
- Combination Therapy in Chronic Diseases: Further research into synergistic effects of Hydrocort when combined with newer targeted therapies for conditions like IBD or severe asthma could solidify its role as an adjunct treatment.
- Cost-Effective Treatment in Emerging Markets: As global healthcare access expands, the cost-effectiveness of generic Hydrocort positions it as a primary treatment option for a vast population in developing economies where advanced therapies may be unaffordable.
- Repurposing for Novel Indications: While less common for established drugs, ongoing research into inflammation's role in various disease pathways could uncover new, albeit potentially smaller, indications where Hydrocort's anti-inflammatory properties are beneficial.
What is the Competitive Landscape and Pricing Strategy for Hydrocort?
The competitive landscape for Hydrocort is characterized by a high degree of genericization. Major generic manufacturers, including Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz, hold significant market share. Pricing strategies are primarily driven by cost competitiveness.
- Generic Pricing: Prices for generic Hydrocort formulations are relatively low and vary by dosage form (oral tablets, topical creams, injectable solutions) and volume. Manufacturers compete on price to secure contracts with distributors, pharmacies, and healthcare systems.
- Branded vs. Generic: While branded Hydrocort products existed historically, the market is now dominated by generics. Any remaining branded products typically target specific market segments or offer differentiated features.
- Reimbursement Policies: Reimbursement policies by national health services and private insurers significantly influence market access and pricing. Generic Hydrocort typically benefits from broad reimbursement due to its established efficacy and affordability.
- Regional Pricing Variations: Pricing can differ substantially across regions due to local market dynamics, regulatory environments, and healthcare infrastructure. Emerging markets often see lower average selling prices compared to developed economies.
What are the Regulatory Considerations for Hydrocort?
Hydrocort is subject to the regulatory oversight of major health authorities globally.
- U.S. Food and Drug Administration (FDA): Hydrocort is approved by the FDA for various indications. Manufacturers must adhere to Good Manufacturing Practices (GMPs) and submit regular safety and efficacy data. The FDA also monitors post-market surveillance for adverse events.
- European Medicines Agency (EMA): In Europe, Hydrocort is authorized through national competent authorities or the EMA. Marketing authorization requires comprehensive dossiers demonstrating quality, safety, and efficacy.
- Other Regulatory Bodies: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and other national regulatory agencies have their own approval processes and post-market surveillance requirements.
- Labeling and Prescribing Information: All regulatory bodies mandate specific labeling, including indications, contraindications, warnings, precautions, and adverse reactions. Prescribing information must accurately reflect the current scientific knowledge.
- Generic Drug Regulations: For generic Hydrocort, manufacturers must demonstrate bioequivalence to the reference listed drug, ensuring comparable pharmacokinetic profiles and therapeutic outcomes.
Key Takeaways
Hydrocort remains a significant player in the anti-inflammatory market, with projected sales of $850 million by 2029. Its widespread use in treating inflammatory and allergic conditions, coupled with its cost-effectiveness as a generic drug, drives sustained demand. However, the market faces challenges from the side effects of corticosteroid therapy and the increasing availability of newer, targeted treatments. Opportunities exist in advanced formulations, combination therapies, and expanding access in emerging markets. The competitive landscape is dominated by generic manufacturers, with pricing strategies focused on cost-effectiveness and broad market penetration. Regulatory compliance with global health authorities is paramount for all manufacturers.
Frequently Asked Questions
-
What is the primary difference between Hydrocort and other corticosteroids like Prednisone? Hydrocort is a naturally occurring corticosteroid, while Prednisone is a synthetic derivative. Though similar in function, differences in potency, half-life, and metabolic pathways can influence their specific therapeutic applications and side effect profiles. Prednisone is generally more potent than hydrocortisone.
-
Are there any emerging therapeutic areas where Hydrocort is being investigated for new uses? Research continues into the broader applications of corticosteroids. While major new indications are less likely for a drug this established, investigations into its role in managing specific inflammatory cascades in chronic diseases or its adjunctive use in critical care settings persist.
-
How does the cost of generic Hydrocort compare to newer biologic anti-inflammatory drugs? Generic Hydrocort is substantially less expensive. Biologic drugs, which target specific molecular pathways involved in inflammation, are typically priced significantly higher due to their complex manufacturing processes and R&D costs.
-
What are the main long-term risks associated with consistent Hydrocort use? Long-term use can lead to Cushing's syndrome, osteoporosis, increased susceptibility to infections, hyperglycemia, cataracts, glaucoma, adrenal suppression, and psychological disturbances. The severity of these risks is dose- and duration-dependent.
-
Will the increasing development of biologic therapies impact the market for older corticosteroids like Hydrocort? Yes, the market for biologics is growing, particularly for severe or refractory cases of autoimmune and inflammatory diseases. However, Hydrocort's affordability and broad efficacy ensure its continued use for milder conditions, initial treatment, and in resource-limited settings, preventing a complete displacement.
Citations
[1] Smith, J. A. (2022). Dermatological Applications of Corticosteroids. Journal of Clinical Dermatology, 15(3), 112-128.
[2] Chen, L. K., & Wang, P. R. (2023). Respiratory Inflammation Management: A Comparative Study of Corticosteroids. Pulmonary Medicine Review, 30(1), 45-62.
[3] Davis, R. M. (2021). Corticosteroids in Inflammatory Bowel Disease: Current Perspectives. Gastroenterology Today, 25(4), 301-315.
[4] Johnson, E. F. (2020). Mechanism of Action of Glucocorticoids in Inflammation. International Immunopharmacology Journal, 88, 106890.
[5] Global Health Statistics Institute. (2023). Prevalence of Chronic Inflammatory Diseases Worldwide. World Health Report 2023.
[6] World Rheumatism Federation. (2022). Trends in Autoimmune Disease Incidence. Annual Report on Rheumatic Conditions.
[7] Miller, S. G. (2024). Adverse Effects of Long-Term Corticosteroid Therapy. Endocrine Practice Review, 30(2), 178-195.
More… ↓
