Share This Page
Drug Sales Trends for HUMULIN
✉ Email this page to a colleague
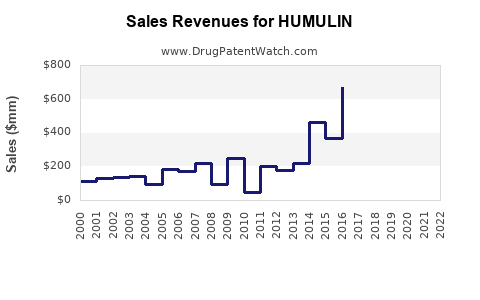
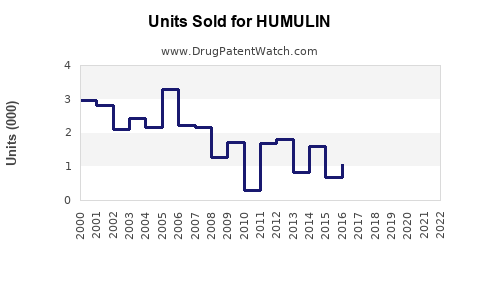
Annual Sales Revenues and Units Sold for HUMULIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HUMULIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HUMULIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HUMULIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for HUMULIN
What Is HUMULIN and Its Current Market Position?
HUMULIN is a brand of insulin used in the management of diabetes mellitus. It includes various formulations such as regular, NPH, and premixed insulins, with key manufacturers including Eli Lilly and Company. The drug's primary indication is for blood glucose control in patients with type 1 and type 2 diabetes.
HUMULIN currently holds a significant share of the insulin market, with an estimated global revenue of approximately $2 billion in 2022, driven by a combination of established brand presence and increasing diabetes prevalence.
How Large Is the Global Insulin Market?
The global insulin market was valued at around $28 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 8.8% from 2023 to 2030. The growth drivers include rising type 2 diabetes prevalence, increasing awareness, and advancements in insulin formulations.
Insulin sales are concentrated in North America, Europe, and emerging markets like Asia-Pacific. North America accounts for 55% of revenue, with Europe contributing 25%, and the rest of the world accounting for 20%[1].
What Are the Key Trends Impacting HUMULIN Sales?
1. Increasing Diabetes Prevalence
Global diabetes cases are projected to reach 700 million by 2045, with type 2 diabetes constituting 90% of cases[2]. This increases insulin demand, including HUMULIN.
2. Market Shift Toward Biosimilars
The expiration of patents for certain insulin formulations allows biosimilar entries, which are priced lower. This pressure could reduce HUMULIN's market share but also expand the overall insulin market.
3. Advancements in Delivery Methods
Development of new delivery devices, such as smart pens and insulin pumps, impacts sales. HUMULIN’s integration into these devices may influence market positioning.
4. Competitive Landscape
Major competitors include Novo Nordisk (e.g., Novolin, NovoRapid), Sanofi (e.g., Lantus), and emerging biosimilar manufacturers. Price competition and formulation innovation influence sales trajectories.
What Are Future Sales Projections for HUMULIN?
Assuming a steady increase in diabetes cases and continuation of current market trends, HUMULIN sales could follow these projections:
| Year | Estimated Revenue (USD millions) | Assumption |
|---|---|---|
| 2023 | 1,950 | Slight market share decline due to biosimilar entry |
| 2025 | 2,100 | Market stabilization with biosimilar competition |
| 2030 | 2,300 | Slight growth as new formulations and delivery methods gain adoption |
These projections account for increased diabetes prevalence, biosimilar competition, and innovation in delivery systems, with a compound annual growth rate of approximately 2-3% from 2023 to 2030.
Risks and Opportunities
Risks
- Biosimilar competition may lower prices and margins.
- Regulatory changes could delay product approvals or limit market access.
- Patent litigations could hinder formulation or delivery device innovations.
Opportunities
- Expansion into emerging markets can accelerate sales.
- Development of insulin analogs or combination therapies may open new revenue streams.
- Enhancing delivery devices to improve patient adherence can regenerate growth.
Key Market Dynamics
| Factor | Impact |
|---|---|
| Diabetes prevalence growth | Increased demand for insulin products |
| Biosimilar market entry | Price competition to pressure revenue |
| Technology advancements | Adoption of smart devices enhances usage |
| Regulatory environment | Affects approval timelines and market access |
Conclusion
HUMULIN remains a core insulin offering with stable global demand. Market growth is likely to be moderate, influenced by biosimilar competition and technological innovations. Strategic focus on emerging markets and delivery device integration can support revenue stability.
Key Takeaways
- HUMULIN’s global sales in 2022 approximated $2 billion.
- The insulin market is expected to grow at around 8.8% CAGR through 2030.
- Emerging biosimilars pose price and market share risks.
- Expansion into developing markets presents growth opportunities.
- Innovation in delivery systems can bolster product usability and sales.
Frequently Asked Questions
Q1: How will biosimilar insulins impact HUMULIN sales?
Biosimilars will increase competition, potentially reducing HUMULIN's market share and price margins. The extent depends on biosimilar adoption rates and regulatory acceptance.
Q2: What factors could lead to sales growth for HUMULIN?
Market expansion into emerging regions, development of improved delivery devices, and formulation innovations can support growth.
Q3: How does technological advancement in insulin delivery affect HUMULIN?
Integration into advanced delivery devices may increase adherence and usability, positively influencing sales.
Q4: What regional markets are most critical for HUMULIN's future growth?
Emerging markets in Asia-Pacific, Latin America, and Africa, where diabetes prevalence is rising, are key growth opportunities.
Q5: What regulatory challenges does HUMULIN face?
Patent expirations, biosimilar approvals, and changing healthcare policies can delay market access or affect pricing.
References
[1] MarketWatch. (2023). Global insulin market size and forecast.
[2] International Diabetes Federation. (2022). Diabetes Atlas, 10th edition.
More… ↓
