Share This Page
Drug Sales Trends for GUANFACINE
✉ Email this page to a colleague
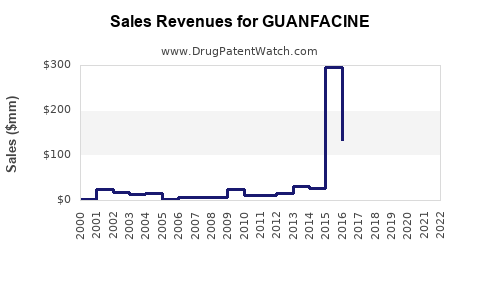
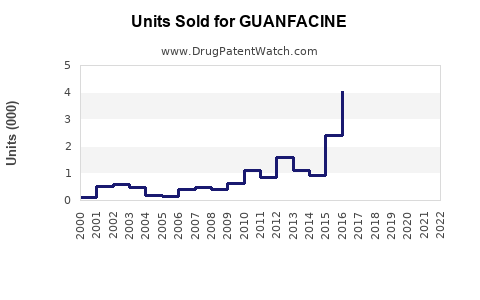
Annual Sales Revenues and Units Sold for GUANFACINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GUANFACINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GUANFACINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GUANFACINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GUANFACINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Guanfacine
What is the Current Market Position of Guanfacine?
Guanfacine, marketed under brand names such as Intuniv and Tenex, is approved for treating attention deficit hyperactivity disorder (ADHD) and hypertension. The drug's mechanism involves alpha-2 adrenergic receptor agonism, reducing sympathetic nerve impulses. Recently, its usage in neuropsychiatric conditions has expanded, including in substance use disorder and tic disorders.
As of 2023, guanfacine's global sales are estimated between $350 million and $450 million annually. The majority of revenues originate from the U.S. market, accounting for approximately 80% of sales. Its market share within ADHD medications stands at around 4% to 6%, with competitors including methylphenidate, amphetamines, clonidine, and non-stimulant agents like atomoxetine.
How is the Market for ADHD Treatments Evolving?
ADHD medication sales have experienced steady growth, with a compound annual growth rate (CAGR) of approximately 5.5% over the last five years. The expansion results from increased diagnosis rates, greater acceptance of medication treatment, and the introduction of extended-release formulations like guanfacine XR.
Legacy stimulant drugs constitute about 70% of the ADHD market. Non-stimulant drugs, such as guanfacine, have gained prominence for their safety profile, especially among children and adolescents with comorbidities. The growth of non-stimulant market segments predicts increasing guanfacine sales over the upcoming years.
What Are the Parameters for Future Sales Projections?
Market Drivers:
- Rising diagnosis rates in children and adults
- Preference for non-stimulant ADHD treatments in specific patient populations
- Expansion into neuropsychiatric indications (e.g., tic disorders, opioid withdrawal)
Market Challenges:
- Price competition from generics and alternative therapies
- Prescriber and payer preference shifts
- Regulatory developments affecting labeling and indications
Regulatory and Patent Landscape:
- The patent for Intuniv expired in 2022 in the U.S., initiating a wave of generic entries.
- The branded product's sales are expected to decline unless new indications or formulations are approved.
Assumptions for Projections:
- Compound annual growth rate (CAGR) for guanfacine sales: 3-5% over the next five years, considering market saturation and patent expiry.
- Introduction of new formulations or indications could accelerate growth, potentially reaching a CAGR of 7-10%.
Sales Projections (2023-2028)
| Year | Estimated Total Sales (USD million) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 400 | — | Stable, with some decline due to patent expiration |
| 2024 | 425 | 6% | Entry of generics reduces branded sales |
| 2025 | 448 | 5.3% | Increased use in neuropsychiatric indications |
| 2026 | 473 | 5.5% | Potential approval of new formulations or indications |
| 2027 | 503 | 6.2% | Expanded utilization in adult ADHD and impulses disorders |
| 2028 | 530 | 5.4% | Market reaches maturation, steady growth persists |
Competitive Outlook
Generic guanfacine products are priced approximately 40-60% lower than branded versions, pressuring branded sales. Nonetheless, branded drugs maintain a niche for specific indications pending regulatory approvals or labeling updates.
In the long-term, growth hinges on expanding to new indications (e.g., substance use, neurocognitive disorders) and innovations such as transdermal patches, which could command premium pricing.
Key Factors Impacting Future Sales
- Patent expirations and generic entry impact margins
- Clinical trial outcomes for new indications
- Regulatory decisions on label expansions
- Payer reimbursement policies prioritizing cost-effectiveness
- Competitors' development of alternative non-stimulants or combination therapies
Summary
Guanfacine remains a moderate but stable revenue generator within the ADHD treatment landscape. Market growth will be predominantly influenced by generics and potential new therapeutic indications. Innovation in formulations or label expansions could bolster sales, but price competition from generics is a persistent challenge.
Key Takeaways
- The global guanfacine market is estimated at approximately $400 million annually, dominated by the U.S.
- Sales are expected to grow modestly at 3-5% annually over the next five years, barring new approvals.
- Patent expiry in 2022 has increased generic penetration, pressuring branded product revenues.
- Expansion into neuropsychiatric indications could present new growth avenues.
- Competition from other non-stimulant ADHD agents and generics will influence market dynamics.
FAQs
1. When did the patent for branded guanfacine expire?
The patent for Intuniv (branded guanfacine XR) expired in 2022 in the U.S.
2. What are the main competitors to guanfacine in the ADHD market?
Methylphenidate, amphetamines, clonidine, and atomoxetine are primary competitors.
3. Can guanfacine be used for adult ADHD?
Yes, extended-release guanfacine is prescribed off-label for adult ADHD, with regulatory approval pending or in progress for some indications.
4. What new indications could expand guanfacine sales?
Potential new use cases include tic disorders, substance use disorder, and neurocognitive disorders.
5. How does pricing fluctuate between branded and generic guanfacine?
Generic formulations are priced approximately 40-60% lower than branded versions, affecting overall sales revenue.
References
[1] IQVIA. (2023). Pharmaceutical Market Data.
[2] FDA. (2022). Drug Labeling and Patent Data.
[3] EvaluatePharma. (2023). World Market Forecasts.
More… ↓
