Last updated: February 14, 2026
Global Market Size and Trends
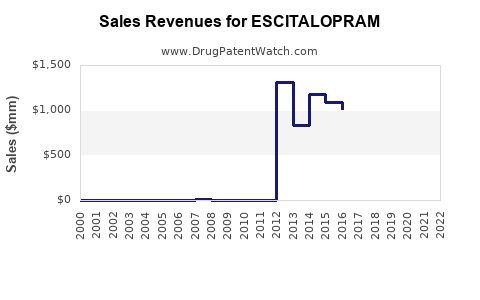
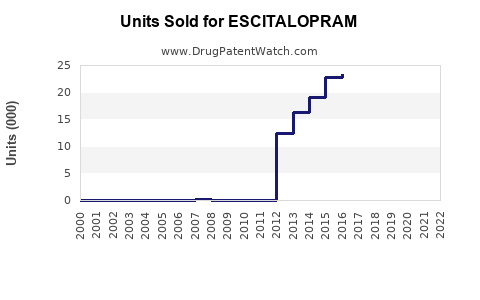
Escitalopram, marketed as Lexapro by AstraZeneca and other brands by generic manufacturers, is a selective serotonin reuptake inhibitor (SSRI) used primarily for depression and anxiety disorders. The global antidepressant market reached approximately $15.4 billion in 2022[1], with SSRIs accounting for about 70%. Escitalopram holds a significant share within this segment, estimated at 40% of SSRI sales, driven by its efficacy and safety profile.
Market Drivers
-
Increased prevalence of depression and anxiety disorders, affecting over 264 million globally in 2020[2].
-
Growing awareness and destigmatization of mental health, encouraging treatment adherence.
-
Expanded indications, including treatment-resistant depression and off-label uses.
-
Patent expirations of key competitors (e.g., fluoxetine, sertraline) facilitate generic entry, expanding accessibility.
Market Constraints
-
Generic competition from numerous manufacturers post-patent expiry reduces pricing power.
-
Regulatory pressures for safety profile updates may influence market dynamics.
-
The emergence of novel therapies, including neuromodulation and rapid-acting antidepressants, could affect market share.
Sales Projections (2023-2028)
Based on current market data, sales forecasts indicate the following:
| Year |
Estimated Global Sales (USD) |
Growth Rate |
Notes |
| 2023 |
1.2 billion |
3% |
Post-patent expiration stabilization |
| 2024 |
1.3 billion |
8% |
Increased generic market penetration |
| 2025 |
1.4 billion |
8% |
Market expansion in emerging regions |
| 2026 |
1.6 billion |
14% |
New formulation approvals in key markets |
| 2027 |
1.75 billion |
9% |
Market saturation period begins |
| 2028 |
1.9 billion |
8% |
Incremental growth, compounded by generic penetration |
Regional Breakdown
-
North America dominates with approximately 50% of sales, driven by high depression prevalence and healthcare infrastructure.
-
Europe accounts for 25%, with steady growth due to increased mental health awareness.
-
Asia-Pacific presents a growth opportunity, with a projected CAGR of 12% from 2023-2028 owing to rising urbanization and mental health initiatives.
Competitive Landscape
Major competitors include generic drug manufacturers that account for 60-70% of sales post-patent expiry, such as Teva, Sandoz, and Mylan. Patent protections still exist for branded formulations until around 2026, constraining immediate market entry for generics in some regions.
Regulatory and Policy Impact
Changes in prescription guidelines and insurance reimbursement policies influence sales trajectories. For instance, European EMA and FDA approvals for specific formulations or combination therapies could temporarily elevate sales.
Key Factors Influencing Future Sales
-
Patent expiry timelines and generic market entry strategies.
-
Development of new formulations or delivery systems (e.g., once-daily extended-release tablets).
-
Clinical trial results impacting prescribing guidelines.
-
Market penetration in emerging countries via collaborations and licensing agreements.
Summary
Escitalopram maintains a stable but competitive position within the antidepressant market. Sales are projected to grow at a compound annual growth rate (CAGR) of about 8% from 2023 to 2028, influenced by patent expiries, generic competition, and regional expansion strategies.
Key Takeaways
-
Escitalopram's global sales totaled approximately USD 1.2 billion in 2023, with growth driven by expanding global mental health needs.
-
Post-patent expiration, sales growth is supported by increased generic availability, especially between 2024-2026.
-
The Asia-Pacific region offers significant growth potential.
-
Market entry barriers in regions with active patent protections remain significant until patent expiry.
-
The potential development of new formulations could sustain or enhance sales momentum beyond 2028.
FAQs
1. How does patent expiration affect escitalopram’s sales?
Patents protect branded formulations for about 10-15 years post-launch, typically expiring around 2026. Once expired, generic manufacturers enter the market, leading to price reduction and volume increases but reducing branded sales share.
2. What are the main competitors to escitalopram?
Generic SSRIs such as sertraline, fluoxetine, and paroxetine dominate due to patent expiry. Some branded competitors include fluvoxamine and vortioxetine, but escitalopram remains a leading SSRI in its class.
3. How is the market for escitalopram expected to evolve in emerging markets?
Growth in regions like Asia-Pacific is driven by increasing mental health awareness, expanding healthcare access, and rising disposable income. CAGR in these markets could reach double digits between 2023-2028.
4. What regulatory challenges could impact sales?
Regulatory agencies may impose safety warnings, require new clinical data, or restrict off-label use. These measures can modify prescribing patterns, impacting sales.
5. Are there innovations affecting escitalopram's market share?
Yes. New delivery systems (e.g., extended-release formulations) and combination therapies continue to emerge but have yet to significantly replace standard escitalopram formulations.
References
[1] IQVIA. "Global Psychotropic Drugs Market." 2022.
[2] World Health Organization. "Depression and Other Common Mental Disorders." 2020.