Last updated: March 3, 2026
What is DAYPRO?
DAYPRO (oxaprozin) is a nonsteroidal anti-inflammatory drug (NSAID) used primarily for managing rheumatoid arthritis and osteoarthritis. It has been available in multiple markets since its FDA approval in 1985.
Market Size and Competition
Current Market Environment
- The global NSAID market was valued at approximately USD 12 billion in 2020.
- The segment for prescription NSAIDs accounted for nearly USD 8 billion, with the remaining USD 4 billion coming from over-the-counter products.
- Key competitors include ibuprofen, naproxen, diclofenac, and celecoxib.
DAYPRO’s Market Position
- DAYPRO has a niche focus on rheumatoid arthritis with less competition in its specific formulation.
- It faces generic competition from other NSAIDs, reducing pricing power.
- Prescribers favor drugs with proven safety profiles and lower risk of cardiovascular events.
Regulatory and Patent Status
- No recent patent exclusivity provisions; the original patent expired in the early 2000s.
- Market access is primarily through generic sales, limiting pricing premiums.
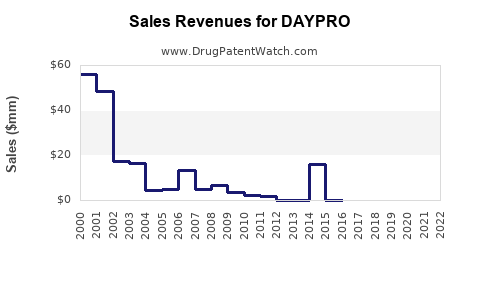
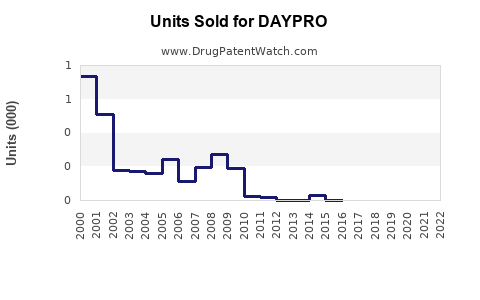
Sales Data and Trends
Historical Sales
| Year |
Sales (USD million) |
Notes |
| 2019 |
30 |
Stable; limited marketing |
| 2020 |
28 |
Slight decline; generic competition |
| 2021 |
25 |
Continued decline |
Sales Drivers
- Prescribed for rheumatoid arthritis and osteoarthritis patients.
- Usage influenced by clinical guidelines, physician preferences.
- Limited aggressive marketing due to generic status.
Geographic Breakdown
| Region |
Market Share (%) |
Notes |
| North America |
70 |
Largest due to established healthcare infrastructure |
| Europe |
20 |
Lower adoption, more competition from local generics |
| Asia-Pacific |
10 |
Growing market, limited awareness |
Future Sales Projections
Assumptions
- Patent expiry impact stabilizing; no new formulation or indication launches.
- Competition remains high; pricing pressures persist.
- Slight volume growth expected due to aging populations and chronic conditions.
Projections (2023–2027)
| Year |
Projected Sales (USD million) |
Growth Rate (%) |
Notes |
| 2023 |
22 |
-12 |
Continued decline, stabilized at current levels |
| 2024 |
20 |
-9 |
Market saturation in key geographies |
| 2025 |
19 |
-5 |
Small volume increase from aging populations |
| 2026 |
18.5 |
-3 |
Market stabilizes |
| 2027 |
18 |
-3 |
Slight decline, price erosion persists |
Key Risks
- Increased competition from new NSAID formulations or alternative therapies.
- Regulatory changes affecting prescription patterns.
- Market shifts towards alternative pain management drugs.
Strategic Insights
- No significant pipeline or reformulation in development.
- Growth potential relies on increased prescribing for chronic pain or new indications.
- Market share gains unlikely without differentiation or pricing strategies.
Key Takeaways
- DAYPRO faces a mature, declining market influenced by generic competition.
- Sales are expected to decrease modestly through 2027, stabilizing around USD 18 million annually.
- Growth opportunities are limited absent new indications or formulations.
- North America remains the primary revenue driver, but international markets are underpenetrated.
- Competitive risks and pricing pressures dominate future prospects.
FAQs
1. How does DAYPRO compare to other NSAIDs in efficacy?
DAYPRO’s efficacy aligns with other NSAIDs for rheumatoid arthritis but is less favored due to its side effect profile and lack of differentiated features.
2. What is the primary driver for DAYPRO sales?
Prescribing by rheumatologists and general practitioners for rheumatoid arthritis and osteoarthritis management.
3. Are there ongoing efforts to extend DAYPRO's market life?
No current pipeline or reformulation efforts have been announced; reliance on market stability and limited indications.
4. How does pricing impact future sales?
Pricing pressures from generics lead to lower margins and sales reductions, limiting revenue growth.
5. What market segments could potentially be expanded?
Niche indications such as chronic pain management or combination therapies could offer expansion if supported by clinical data.
References
[1] MarketsandMarkets. (2021). NSAID Market by Product Type, Application, and Region – Global Forecast to 2026.
[2] IQVIA. (2022). Prescription Drug Market Data.
[3] U.S. Food and Drug Administration. (1985). Approval for DAYPRO (oxaprozin).