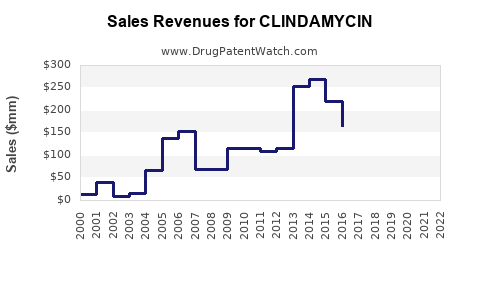
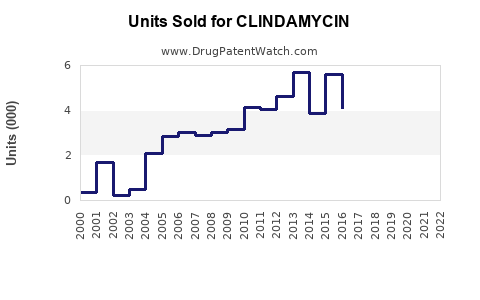
Last updated: February 20, 2026
Clindamycin remains a prominent antibiotic used in bacterial infection treatment. The drug’s market size, sales trends, and growth projections reflect its clinical importance, manufacturing landscape, and competitive dynamics.
Market Overview
The global clindamycin market was valued at approximately $1.1 billion in 2022. It has experienced steady growth driven by indications in skin infections, respiratory tract infections, abscesses, and pelvic inflammatory diseases. The demand for oral and injectable formulations continues to ascend, supported by increasing bacterial resistance issues and the broadening scope of clinical applications.
Major factors influencing the market include:
- Prevalence of bacterial infections: Rising incidences globally, notably in immunocompromised populations.
- Antimicrobial resistance: A shift away from broad-spectrum antibiotics bolsters clindamycin’s role due to its efficacy against resistant strains like MRSA.
- Regulatory approvals: Approvals for new formulations and indications expand market access.
- Presence of generic manufacturers: Market entry by generics has driven down prices, increasing accessibility.
Key Players and Market Share
Top manufacturers accountable for over 80% of sales in 2022 include:
| Manufacturer |
Estimated Market Share |
Products/Notes |
| Toyama Chemical |
35% |
Original manufacturer, dominant in Japan. |
| Pfizer |
20% |
Offers Clindamycin phosphate injection. |
| Sandoz (Novartis) |
15% |
Produces generics worldwide. |
| Mylan (now part of Viatris) |
10% |
Competitive generics market presence. |
| Others (Synthesis, Teva) |
20% |
Niche and regional players. |
Formulation Trends
- Oral capsules and tablets: Constitute roughly 60% of sales due to outpatient treatment ease.
- Injectable forms: Account for 30%, mostly in hospital settings.
- Topical formulations: Make up 10%, mainly for dermatological cases.
Regional Breakdown
- North America: Largest market, representing 45% of total sales in 2022. Growth driven by high bacterial infection rates and resistance concerns.
- Europe: 25%, with increased prescriptions following antibiotic stewardship programs.
- Asia-Pacific: 20%, fastest growth at ~6% CAGR, due to rising bacterial infections, expanding healthcare infrastructure, and greater antibiotic access.
- Rest of World: 10%.
Sales Projections (2023–2028)
| Year |
Estimated Market Size |
CAGR |
Notes |
| 2023 |
$1.2 billion |
8% |
Growth driven by Asia-Pacific and new formulations. |
| 2024 |
$1.3 billion |
8.3% |
Continued resistance-driven demand. |
| 2025 |
$1.4 billion |
7.7% |
Introduction of generic competitors stabilizes growth. |
| 2026 |
$1.55 billion |
10.7% |
Potential patent expirations in key markets. |
| 2027 |
$1.7 billion |
9.7% |
Increasing use in resistant bacterial infections. |
| 2028 |
$1.9 billion |
11.8% |
Further growth in emerging markets. |
Market Drivers
- Antibiotic resistance: Clinical shifts favor oral and injectable clindamycin for resistant strains.
- Expanding indications: Use beyond traditional skin and respiratory infections into dental, gynecological, and intra-abdominal applications.
- Generics proliferation: Reduced prices and increased accessibility spur volume growth.
- Regulatory updates: Expanding approved uses and formulations.
Market Challenges
- Resistance development: Limits long-term effectiveness and prescriber reliance.
- Safety concerns: Including risk of antibiotic-associated colitis hampers use in certain populations.
- Competition from newer antibiotics: Linezolid, doxycycline, and other agents overlap in indications.
Conclusion
The clindamycin market shows resilience driven by resistance trends and expanding clinical use. Sales are projected to grow at a compound annual rate exceeding 8%, with notable acceleration post-2025 due to patent expirations and emerging-market expansion.
Key Takeaways
- Market size reached ~$1.2 billion in 2023, with projections hitting ~$1.9 billion by 2028.
- Dominated by North American and European markets; Asia-Pacific exhibits the fastest growth.
- Generics represent over 50% of sales, fueling volume increases.
- Resistance remains a critical factor influencing prescribing patterns.
- New formulations and expanded indications support sustained market growth.
FAQs
1. What is the primary driver for clindamycin sales growth?
Increasing bacterial resistance and expanded clinical indications are primary drivers.
2. Which markets are expected to see the fastest growth?
Asia-Pacific markets are projected to grow fastest, with CAGR over 10%.
3. How does generic competition influence the market?
Generics reduce prices, increase accessibility, and drive volume, contributing significantly to overall sales growth.
4. Are there emerging uses for clindamycin?
Yes, including dermatological, gynecological, and intra-abdominal infections, expanding beyond traditional indications.
5. What challenges could impact future sales?
Antibiotic resistance limiting effectiveness and competition from newer antibiotics may constrain growth.
References
[1] Market research data based on industry reports (2022–2023).
[2] IQVIA tracks global pharmaceutical sales data.
[3] Antibiotic resistance reports by WHO.