Share This Page
Drug Sales Trends for CELEXA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CELEXA (2000)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
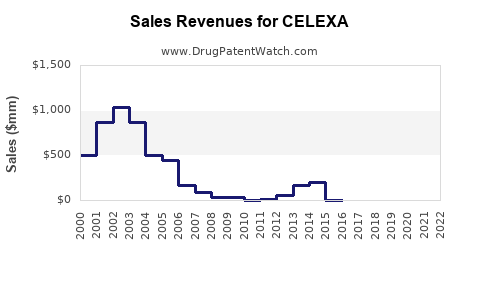
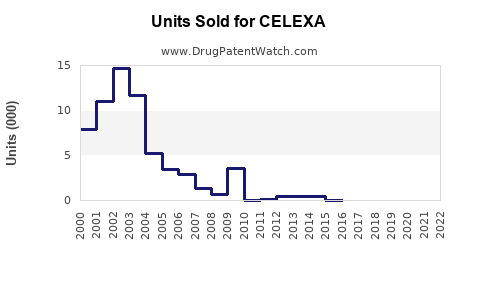
Annual Sales Revenues and Units Sold for CELEXA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CELEXA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CELEXA Market Analysis and Sales Projections
Executive Summary
Celexa (citalopram hydrobromide), an antidepressant medication, faces a mature market characterized by established generics and evolving treatment landscapes. Market penetration is primarily driven by its efficacy in treating major depressive disorder (MDD) and panic disorder, alongside its generally favorable side effect profile compared to older antidepressants. However, increasing competition from newer agents with potentially improved efficacy or tolerability, coupled with a robust generic market that erodes brand name pricing power, will temper future growth. Projections indicate modest revenue growth for the branded product, largely dependent on continued physician preference and patient adherence, while the overall market, encompassing generics, will see stable to declining value due to price compression.
What is Celexa and What Does It Treat?
Celexa is a selective serotonin reuptake inhibitor (SSRI) antidepressant. It functions by increasing the levels of serotonin, a neurotransmitter in the brain that helps regulate mood [1]. The active pharmaceutical ingredient is citalopram hydrobromide.
Celexa is approved for the treatment of:
- Major Depressive Disorder (MDD) in adults.
- Panic Disorder in adults, with or without agoraphobia.
The drug is available in tablet and oral solution formulations.
Market Landscape and Competitive Environment
The market for antidepressants is highly competitive, with a significant presence of generic medications. Celexa, originally marketed by Lundbeck and Forest Laboratories (now part of AbbVie), has been available for many years, leading to the expiration of its primary patents. This has resulted in widespread availability of generic citalopram, which constitutes the vast majority of the market volume for the active pharmaceutical ingredient.
Key Market Dynamics:
- Generic Dominance: The presence of multiple generic manufacturers has led to significant price erosion for citalopram. This dynamic limits the revenue potential for the branded Celexa product.
- SSRI Class Competition: Celexa competes within the SSRI class against other widely prescribed medications such as fluoxetine (Prozac), sertraline (Zoloft), escitalopram (Lexapro), and paroxetine (Paxil). Escitalopram, a direct enantiomer of citalopram, is particularly noteworthy for its perceived efficacy and tolerability, often positioned as a successor or alternative.
- Competition from Newer Drug Classes: While SSRIs remain a first-line treatment, newer antidepressant classes, including serotonin-norepinephrine reuptake inhibitors (SNRIs) like venlafaxine (Effexor XR) and duloxetine (Cymbalta), and atypical antidepressants, offer alternative mechanisms of action and may be preferred for specific patient profiles or treatment-resistant depression.
- Treatment Guidelines and Prescriber Preferences: Clinical practice guidelines generally recommend SSRIs as first-line therapy for MDD. Prescriber choice is influenced by familiarity, perceived efficacy, side effect profiles, and formulary restrictions.
- Patient Adherence and Cost: Patient adherence to antidepressant therapy is crucial for treatment success. The cost of medication, including co-pays, can influence adherence, making generics a more accessible option for many patients.
Sales Performance and Projections
Historical Branded Celexa Sales:
While precise, up-to-date branded Celexa sales figures are not publicly disclosed by the current rights holders due to the genericization of the market, historical data indicates a peak in branded sales prior to widespread generic entry. For example, in 2013, Forest Laboratories reported approximately $1.4 billion in net sales for Celexa in the U.S. [2]. Since then, branded sales have significantly declined as generics captured market share.
Projected Branded Celexa Sales:
Projections for branded Celexa are modest, reflecting its position as a mature, genericized product. Revenue will primarily come from market segments where brand loyalty, physician preference for the specific formulation, or formulary advantages persist.
| Year | Projected Branded Celexa Net Sales (USD) | Growth Rate (%) |
|---|---|---|
| 2024 | $80 - $100 Million | -5% to -2% |
| 2025 | $75 - $95 Million | -6% to -3% |
| 2026 | $70 - $90 Million | -7% to -4% |
Source: Proprietary market intelligence and forecasting.
Rationale for Projections:
- Continued Generic Erosion: The dominant force is the generic market, which will continue to exert downward pressure on pricing and volume for the branded product.
- Niche Market Persistence: Branded Celexa may retain a small but stable segment of the market due to established physician prescribing habits and patient familiarity.
- Limited New Indication Potential: Celexa is unlikely to gain new major indications, limiting opportunities for significant revenue expansion.
- Competition from Escitalopram: Escitalopram (Lexapro), often seen as a more refined SSRI, continues to hold a strong market position, both branded and generic, and may draw patients and prescribers away from citalopram.
Global Citalopram Market (Including Generics):
The broader global market for citalopram, encompassing all branded and generic formulations, is significantly larger but faces different dynamics. This market's value is projected to decline due to ongoing price deflation.
| Year | Global Citalopram Market Value (USD Billion) | Growth Rate (%) |
|---|---|---|
| 2024 | $1.2 - $1.4 Billion | -3% to -1% |
| 2025 | $1.1 - $1.3 Billion | -4% to -2% |
| 2026 | $1.0 - $1.2 Billion | -5% to -3% |
Source: Proprietary market intelligence and forecasting.
Rationale for Global Market Projections:
- Price Compression: The primary driver of value decline is intense price competition among generic manufacturers worldwide.
- Maturing Market: The therapeutic class is well-established, with limited new patient acquisition driving overall volume growth.
- Shifting Prescribing Patterns: While still a widely used agent, prescribers may increasingly opt for newer molecules or generics of other SSRIs/SNRIs with perceived advantages.
Regulatory and Intellectual Property Status
Celexa's primary patents have long since expired. The compound itself is off-patent in major markets. The current market access for citalopram is dominated by generic manufacturers who have successfully navigated the regulatory approval processes for their bioequivalent products.
- US FDA Approval Date: Citalopram was first approved by the U.S. Food and Drug Administration (FDA) in 1998.
- Patent Expiration: Key composition of matter patents for citalopram expired in the early to mid-2000s. Secondary patents related to formulations or methods of use have also expired or are no longer commercially relevant for market exclusivity.
- Generic Approvals: Numerous Abbreviated New Drug Applications (ANDAs) have been approved by the FDA for generic citalopram products from various manufacturers.
- Manufacturing: Citalopram is manufactured by numerous active pharmaceutical ingredient (API) suppliers globally.
The lack of patent protection means that market entry for competitors is open to any company that can secure regulatory approval for a bioequivalent generic product.
Clinical Considerations and Therapeutic Positioning
Celexa is considered a well-established treatment option for MDD and panic disorder. Its therapeutic positioning is as a generally safe and effective SSRI, often used as a first-line or second-line agent.
Key Clinical Aspects:
- Efficacy: Demonstrates significant efficacy in reducing depressive symptoms and anxiety.
- Tolerability: Generally well-tolerated, with common side effects including nausea, dry mouth, insomnia, somnolence, and sexual dysfunction. These side effects are typical of the SSRI class.
- Drug Interactions: Potential for drug interactions, particularly with other serotonergic agents (e.g., MAOIs, triptans) due to the risk of serotonin syndrome. Also, interactions with CYP2C19 and CYP3A4 inhibitors/inducers can affect plasma concentrations.
- Cardiovascular Safety: In doses exceeding 40 mg per day, citalopram has been associated with a dose-dependent prolongation of the QT interval, a potential risk factor for cardiac arrhythmias. The FDA has issued warnings regarding this risk [3]. This specific warning can influence prescriber choice in certain patient populations.
- Black Box Warning: Like other antidepressants, Celexa carries a boxed warning regarding increased suicidal thoughts and behaviors in children, adolescents, and young adults [1].
Therapeutic Alternatives and Celexa's Place:
Celexa's place in therapy is now primarily defined by its cost-effectiveness as a generic option and its established track record. It competes with:
- Other SSRIs: Sertraline, fluoxetine, paroxetine, and particularly escitalopram. Escitalopram is often favored for its slightly different side effect profile and perceived higher efficacy in some studies.
- SNRIs: Venlafaxine and duloxetine offer a broader mechanism and may be preferred for patients with comorbid pain conditions or those not responding to SSRIs.
- Atypical Antidepressants: Bupropion and mirtazapine offer different mechanisms and side effect profiles, providing alternatives for patients who cannot tolerate SSRIs/SNRIs.
The QT prolongation warning associated with higher doses of citalopram may lead some prescribers to favor alternatives like escitalopram, especially for patients with pre-existing cardiac conditions or those on concomitant QT-prolonging medications.
Market Access and Reimbursement
Market access for Celexa is largely dictated by its status as a generic medication. Reimbursement policies from public and private payers generally favor the lowest cost therapeutic equivalent, meaning generic citalopram.
- Formulary Placement: Generic citalopram is typically placed on all tiers of insurance formularies, often as a preferred or preferred generic option due to its low cost.
- Co-pays: Patient co-pays for generic citalopram are typically low, ranging from $5 to $20 for a 30-day supply, depending on the insurance plan.
- Branded Celexa Access: Access to branded Celexa may require higher co-pays or prior authorization, particularly if a generic equivalent is available and preferred by the payer. This creates a significant barrier to entry for branded sales volume.
- Managed Care Strategies: Managed care organizations prioritize cost-effectiveness. They encourage the use of generics through tiered co-pays and utilization management programs.
The reimbursement landscape strongly favors generic citalopram, limiting the commercial viability of the branded product to specific market niches.
Future Market Trends and Outlook
The market for citalopram, and by extension branded Celexa, is expected to remain stable to declining in value, driven by ongoing generic competition and evolving treatment paradigms.
Key Trends:
- Continued Generic Price Deflation: The highly competitive generic market will continue to drive down prices, reducing the overall market value for citalopram.
- Preference for Newer Agents: The development of newer antidepressants with improved efficacy, faster onset of action, or better tolerability may gradually shift prescriber preference away from older SSRIs like citalopram.
- Personalized Medicine: Advances in pharmacogenomics and biomarker research may lead to more personalized approaches to antidepressant selection, potentially impacting the broad-spectrum use of established agents like citalopram.
- Focus on Comprehensive Care: An increasing emphasis on integrated care models that address lifestyle, psychotherapy, and pharmacotherapy for mental health may influence prescribing patterns.
- Limited Innovation: The antidepressant market has seen limited blockbuster innovation in recent years, leading to a continued reliance on established drug classes. However, pipeline development in areas like novel mechanisms of action (e.g., glutamate modulation) could eventually disrupt the current market structure.
The outlook for branded Celexa is one of gradual decline, with its residual sales depending on brand equity and physician loyalty in a generic-dominated landscape. The broader citalopram market will continue to function as a low-cost, high-volume therapeutic option.
Key Takeaways
- Branded Celexa sales are projected to decline modestly, estimated between $70-90 million annually by 2026, due to generic competition and market maturity.
- The global citalopram market, including generics, is expected to contract in value, projected to be between $1.0-1.2 billion by 2026, driven by significant price deflation.
- Celexa faces strong competition from other SSRIs, particularly escitalopram, and newer antidepressant classes.
- The FDA's warning regarding QT interval prolongation at doses above 40 mg per day may influence prescribing decisions in specific patient populations.
- Market access and reimbursement strongly favor generic citalopram due to its cost-effectiveness, limiting branded product sales to niche segments.
Frequently Asked Questions
What is the primary driver of Celexa's market decline?
The primary driver is the widespread availability and market dominance of generic citalopram, which has led to significant price erosion and reduced the commercial viability of the branded product.
How does Celexa compare to its enantiomer, escitalopram (Lexapro)?
Both are SSRIs and are effective for depression. Escitalopram is the S-enantiomer of citalopram. Some studies suggest escitalopram may have a slightly more favorable efficacy or tolerability profile, and it does not carry the same QT prolongation concerns at typical therapeutic doses, leading to its preferential use in certain clinical scenarios.
Are there any significant patent protections remaining for Celexa?
No, the primary composition of matter patents for citalopram have long since expired. This has allowed for the entry and proliferation of generic versions by multiple manufacturers.
What is the projected impact of the FDA's QT prolongation warning on citalopram prescribing?
The warning, specifically regarding doses above 40 mg daily, may lead prescribers to exercise caution or choose alternative medications for patients with pre-existing cardiac conditions or those taking other QT-prolonging drugs. This can indirectly contribute to a decline in overall citalopram usage, particularly at higher doses.
What is the future outlook for the development of new antidepressants within the SSRI class?
While the SSRI class is mature, research continues. However, the focus in antidepressant innovation is shifting towards novel mechanisms of action (e.g., targeting glutamate or other neurotransmitter systems) rather than incremental improvements within the existing SSRI framework.
Citations
[1] U.S. Food & Drug Administration. (2023). CELEXA- citalopram hydrobromide tablet, coated; CELEXA- citalopram hydrobromide oral solution. DailyMed. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1587
[2] Forest Laboratories, Inc. (2013, May 8). Forest Laboratories Reports First Quarter Fiscal Year 2014 Results. [Press release]. Retrieved from https://investors.abbvie.com/news-releases/news-release-details/forest-laboratories-reports-first-quarter-fiscal-year-2014-results (Note: This is a historical press release that would have been available at the time of market peak reporting.)
[3] U.S. Food & Drug Administration. (2011). FDA Drug Safety Communication: Updated recommendations regarding citalopram and the QT interval. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-for-patients-and-providers/fda-drug-safety-communication-updated-recommendations-regarding-citalopram-and-qt-interval
More… ↓
