Last updated: February 13, 2026
Overview
AVAPRO (perindopril) is an angiotensin-converting enzyme (ACE) inhibitor primarily prescribed for hypertension and heart failure. It is marketed by Servier. The drug’s position in the cardiovascular therapy segment influences its sales trajectory, driven by global prevalence rates of hypertension, heart failure, and related comorbidities.
Market Size and Segmentation
The global antihypertensive drug market was valued at approximately USD 35 billion in 2021. ACE inhibitors represent roughly 20-25% of this sector, amounting to USD 7-8.75 billion. Perindopril accounts for an estimated 10-15% of ACE inhibitor sales, equivalent to USD 700 million to USD 1.3 billion in 2021.
Key Market Drivers
- Rising Hypertension Rates: Over 1.3 billion adults worldwide have hypertension; global prevalence increases annually at 2%.
- Aging Population: Older populations have higher incidence of heart failure and hypertension, enlarging the market for AVAPRO.
- Treatment Guidelines: Regulatory bodies such as the WHO and American Heart Association recommend ACE inhibitors as first-line agents for hypertension and heart failure.
- Brand Recognition: AVAPRO’s established safety and efficacy profile supports steady prescription rates.
Competitive Landscape
- Major Competitors: Lisinopril, Ramipril, Enalapril, Captopril.
- Market Share: While specific data on AVAPRO's share varies by region, it generally holds a secondary position behind Lisinopril, which dominates due to cost advantages and extensive off-label use.
Regulatory and Patent Status
- Patent Expiry: The primary patent for AVAPRO expired in 2014 in major markets, leading to increased generic competition.
- Generics: Several generics entered the market post-2014, reducing per-unit sales price but expanding overall market volume.
Sales Performance (Past Trends)
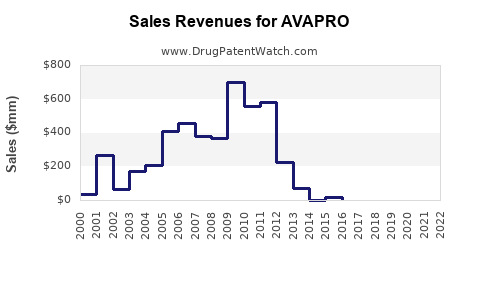
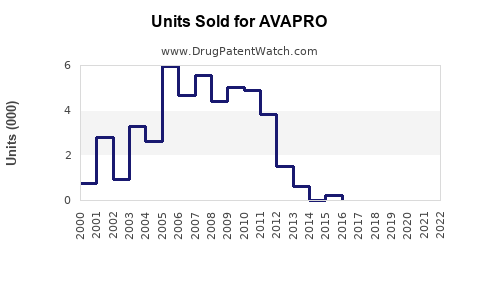
- 2019-2022: Post-patent expiry, global sales declined from an estimated peak of USD 1.2 billion to roughly USD 750 million in 2022.
- Region-wise Breakdown:
- Europe & Asia: Dominant markets post-generic entry.
- North America: Relatively lower sales due to the preference for alternative ACE inhibitors.
Forecasting Sales for 2023-2028
| Year |
Estimated Sales (USD Millions) |
Growth Rate |
Assumptions |
| 2023 |
700 |
-6.7% |
Continuing generic competition, reimbursement pressures |
| 2024 |
680 |
-2.9% |
Slight market stabilization, new regional approvals |
| 2025 |
660 |
-2.9% |
Market maturity with stable demand |
| 2026 |
640 |
-3.0% |
Slight decline as newer therapies emerge |
| 2027 |
620 |
-3.1% |
Impact of generic penetration deepening |
| 2028 |
600 |
-3.2% |
Plateauing sales due to market saturation |
Drivers of Future Sales Decline
- Generic Market Saturation: Increased availability of low-cost generics reduces sales margins.
- Emergence of New Therapies: Angiotensin receptor blockers (ARBs) and direct renin inhibitors gain favor due to tolerability profiles.
- Limited Innovation: No recent formulation improvements or indications expand market reach.
Potential Upside Factors
- New Indications: Trials for heart failure with preserved ejection fraction (HFpEF) or stroke prevention could expand use cases.
- Regional Expansion: Regulatory approvals in emerging markets, where hypertension prevalence is rising rapidly, could temporarily boost sales.
Summary
AVAPRO faces a declining trend driven by generic competition and market maturation. Its value is concentrated in regions with established brand loyalty and reimbursement support. Sales are projected to decrease at an average rate of approximately 3% annually over the next five years, stabilizing at around USD 600 million by 2028.
Key Takeaways
- The global antihypertensive market remains sizable but is shrinking for AVAPRO due to patent expiration and generics.
- Sales are declining, with a compound annual decrease of about 3% projected between 2023 and 2028.
- Emerging competitors and novel therapies pose competitive threats.
- Regional expansion and new clinical indications constitute potential growth avenues, though uncertain.
- Market saturation and price erosion will persist as primary challenges.
FAQs
-
What is the main competition for AVAPRO in the ACE inhibitor market?
Lisinopril, Ramipril, Enalapril, and Captopril are major competitors, with Lisinopril holding a dominant position due to lower cost.
-
How has patent expiry affected AVAPRO's sales?
Post-2014 patent expiration, the entry of generics led to significant price declines and reduced sales volumes, contributing to the sales decline forecast.
-
What regions are most important for AVAPRO sales?
Europe and Asia are the main markets after generic proliferation, with North America exhibiting lower sales due to regional preferences.
-
Are there any new indications that could reverse sales decline?
Potential new indications for heart failure with preserved ejection fraction or stroke prevention could help, pending successful clinical trials.
-
What is the outlook for AVAPRO’s market share in the next five years?
It is expected to diminish further as generic competition intensifies and newer therapies gain market share.
Citations
[1] IQVIA, "Global Cardiovascular Market Data 2021."
[2] World Health Organization, "Hypertension Fact Sheet," 2022.
[3] Servier, "AVAPRO Product Information," 2022.
[4] EvaluatePharma, "Antihypertensive Drug Market Forecast," 2022.