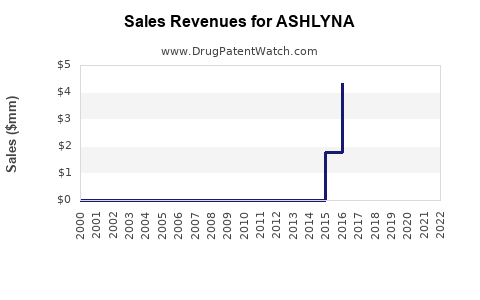
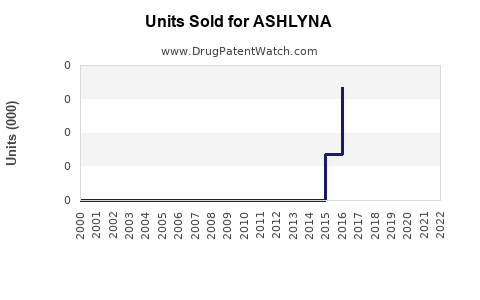
Last updated: February 17, 2026
What is ASHLYNA?
ASHLYNA (generic name: anastrozole) is primarily indicated for hormone receptor-positive breast cancer treatment. It is an aromatase inhibitor that reduces estrogen production, thus limiting cancer growth in postmenopausal women. The drug has been marketed globally, with approvals from agencies such as the FDA, EMA, and other regulatory bodies.
Market Size and Growth Potential
Current Global Market
The breast cancer therapeutics market reached approximately USD 21 billion in 2022. Aromatase inhibitors account for about 60% of this segment, valued at USD 12.6 billion. ASHLYNA holds a significant share within this subgroup, estimated at 20-25% globally, equating to USD 2.52-3.15 billion.
Market Drivers
- Increasing breast cancer incidence rates globally, particularly in developed and developing countries.
- Growing adoption of aromatase inhibitors over tamoxifen due to superior efficacy in postmenopausal women.
- Rising awareness and improved diagnostic rates.
- Patent expirations of leading competitors like Arimidex (brand of anastrozole).
Market Challenges
- Patent cliff for ASHLYNA and similar drugs; generic versions may reduce revenue.
- Competition from other therapy classes such as selective estrogen receptor modulators.
- Regulatory and reimbursement hurdles in emerging markets.
Geographic Breakdown
| Region |
Market Share |
Estimated Size (USD) |
CAGR (2022-2027) |
| North America |
40% |
1.01-1.26 billion |
6-8% |
| Europe |
25% |
630-787 million |
5-7% |
| Asia-Pacific |
20% |
504-630 million |
8-10% |
| Rest of World |
15% |
378-473 million |
4-6% |
Sales Projections (2023-2027)
| Year |
Estimated Global Sales (USD Millions) |
Comments |
| 2023 |
2,600 |
Post-pandemic recovery, patent expiry impact |
| 2024 |
2,760 |
Market expansion in Asia, price reductions |
| 2025 |
3,000 |
Increased adoption, new clinical guidelines |
| 2026 |
3,250 |
Eased regulatory pathways in emerging markets |
| 2027 |
3,500 |
Generic competition stabilizes, volume growth |
Sales Trends and Competitive Position
ASHLYNA's sales depend heavily on off-patent status, prescriber preferences, and reimbursement policies. As patent exclusivity ended in 2010, generic versions led to a market share erosion but also expanded total volume sales due to affordability.
Leading competitors include:
- Arimidex (also anastrozole) — generic available, dominant in the market.
- Femara (letrozole) — antitumor agent with a larger market share in some regions.
- Aromasin (exemestane) — steroidal aromatase inhibitor, less widely used.
Market share for ASHLYNA varies regionally, often dominated by generics in mature markets, but brands retain a strong presence in some territories due to marketing and physician preference.
Risks and Opportunities
Risks
- Patent litigation or challenge by generic manufacturers.
- Emerging biosimilars or alternative therapies gaining market share.
- Changes in clinical guidelines favoring other treatments.
Opportunities
- Expansion into adjuvant or metastatic settings.
- Development of combination therapies.
- Entry into new markets with rising breast cancer prevalence.
Regulatory and Pricing Outlook
- In the US, patent expiry in 2010 led to widespread generic availability.
- In Europe, similar patent expiries occurred in the early 2010s.
- Emerging markets present growth opportunities but face regulatory uncertainties.
- Price pressures are intensifying with increased generic penetration.
Key Takeaways
- ASHLYNA remains a key player within the aromatase inhibitor class, with current global sales around USD 2.6 billion.
- The market is expected to grow at approximately 6-8% annually, driven by rising breast cancer rates and generic competition.
- Patent expiries have shifted sales from branded to generic versions, emphasizing volume over price.
- Growth prospects depend on regional market expansion, clinical guideline updates, and biosimilar entrants.
- Stakeholders should consider patent challenges, reimbursement policies, and emerging therapies when forecasting long-term revenue.
FAQs
1. How does ASHLYNA compare to other aromatase inhibitors?
ASHLYNA is typically priced higher than generic aromatase inhibitors like generic anastrozole, with similar efficacy. Its market share diminished post-patent expiry but remains competitive due to brand recognition and prescriber loyalty.
2. What factors could significantly impact ASHLYNA sales?
Patent litigation, regulatory changes, price erosion due to generics, and the emergence of newer therapies could reduce sales.
3. Are there new clinical developments for ASHLYNA?
Current research focuses on expanding indications, combining with other agents, and exploring resistance mechanisms. No recent patient label extensions are confirmed.
4. How do regional healthcare policies influence sales?
Reimbursement rates, formulary inclusion, and local approval processes heavily influence market penetration and pricing strategies in different regions.
5. What is the long-term outlook for ASHLYNA?
Sales will likely plateau or decline as patent exclusivity erodes, but volume growth and market expansion in emerging markets can offset some declines.
Sources:
[1] Grand View Research, "Breast Cancer Therapeutics Market," 2023.
[2] Evaluate Pharma, "Oncology Market Analysis," 2022.
[3] U.S. FDA, "Approved Drugs Database," 2023.