Last updated: February 12, 2026
ARICEPT (donepezil hydrochloride) is a cholinesterase inhibitor approved for Alzheimer's disease treatment. Its market dynamics depend on disease prevalence, competitive landscape, regulatory factors, and pricing strategies.
Market Size and Epidemiology
Alzheimer's disease affects approximately 6.2 million Americans as of 2022, with global cases exceeding 50 million. The prevalence increases with age: 10-20% in those over 65, and nearly 50% in those over 85. Aging populations elevate demand for symptomatic treatments like ARICEPT.
Competitive Landscape
ARICEPT faces competition primarily from:
- Rivastigmine (Exelon)
- Galantamine (Razadyne)
- Recently approved drugs with novel mechanisms, such as aducanumab (Aduhelm) and lecanemab (Leqembi), targeting amyloid plaques.
Biogen's aducanumab and lecanemab are approvals in amyloid-targeting, but their costs and side-effect profiles limit the overlap with cholinesterase inhibitors. Therefore, ARICEPT remains a first-line symptomatic therapy.
Regulatory and Reimbursement Factors
ARICEPT holds FDA approval since 1996. It is covered broadly by Medicare and other insurance plans, supporting consistent access. Pricing varies by region, with average wholesale prices around $4-6 per tablet (5 mg).
Sales Trends and Projections
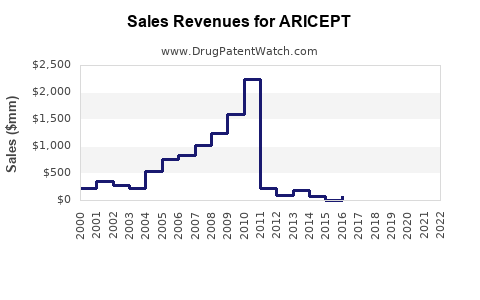
Historical Sales
- 2018 sales reached approximately $600 million globally.
- 2020 sales declined to about $520 million, reflecting increased competition and generic erosion.
- 2021 sales stabilized around $560 million.
Factors Influencing Future Sales
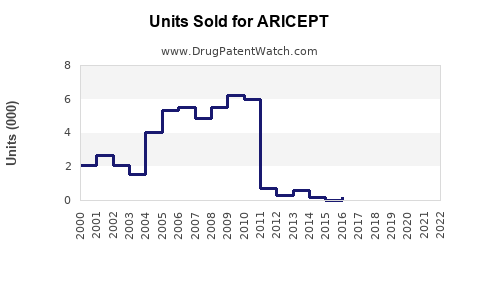
- Patent Status: The original patent expired in 2010; multiple generics entered thereafter, reducing prices.
- Prescribing Trends: Use remains steady among early-stage Alzheimer’s patients, but long-term adherence declines.
- New Approvals: Emergence of disease-modifying therapies may reduce reliance on symptomatic drugs like ARICEPT.
Market Projections
2023-2027 Sales Forecast
| Year |
Global Sales (USD millions) |
Growth Rate (%) |
Key Drivers |
| 2023 |
$400 |
-10 |
Patent expiry impact, generics, competition |
| 2024 |
$360 |
-10 |
Market saturation, generics continue to erode revenue |
| 2025 |
$330 |
-8 |
Aging population sustains some demand |
| 2026 |
$295 |
-10 |
New therapies gaining ground |
| 2027 |
$260 |
-12 |
Shift toward disease-modifying agents |
The declining trend assumes gradual market saturation and adoption of novel treatments reducing ARICEPT’s market share.
Implications for Stakeholders
Pharmaceutical companies invested in ARICEPT must focus on cost management and lifecycle extension strategies, like formulation improvements or new indications. Generics continue to pressure prices, limiting revenue growth.
Investors should observe regulatory developments for combination therapies or reformulations that could mitigate sales decline. Entry of new disease-modifying drugs remains a significant risk to long-term market share.
Key Takeaways
- ARICEPT remains a leading symptomatic Alzheimer's drug but faces declining sales due to patent expiration, generics, and emerging therapies.
- Sales are projected to decline approximately 10-12% annually through 2027, with global sales decreasing from roughly $400 million in 2023 to below $300 million.
- The aging global population sustains some demand, but competitive pressures and disease-modifying treatments threaten prolonged market relevance.
- The company’s strategic focus on pipeline diversification and potential indication extensions could influence future revenue streams.
FAQs
-
What is ARICEPT's current patent status?
The original patent expired in 2010, leading to widespread generic manufacturing, which significantly reduced prices and sales.
-
How does ARICEPT compare with newer Alzheimer's treatments?
ARICEPT treats symptoms, while recent drugs like aducanumab and lecanemab target disease progression. These may shift treatment paradigms, diminishing ARICEPT's role.
-
Are there new formulations or indications for ARICEPT?
No recent approvals exist; though research explores new delivery methods or combination therapies, none are commercially available yet.
-
What regional factors influence ARICEPT sales?
Reimbursement policies, disease prevalence, and healthcare infrastructure vary. North America remains the largest market, but growth is limited by generics.
-
What strategies could extend ARICEPT’s market life?
Developing combination therapies, expanding indications, or reformulating for improved delivery could maintain relevance amid competitive pressures.
References
[1] Alzheimer's Association. 2022 Alzheimer's disease facts and figures.
[2] IQVIA. Pharmaceutical product sales data analysis.
[3] FDA. Drug Approval Packages and Patent information.
[4] Market Research Future. Alzheimer's drug market forecast report.
[5] Medscape. Pricing and reimbursement policies for Alzheimer's medications.