Last updated: February 13, 2026
APRISO (mesalamine) is a delayed-release formulation of 5-aminosalicylic acid (5-ASA), primarily approved for the treatment of ulcerative colitis (UC) in adults. Its targeted delivery to the colon improves local efficacy while minimizing systemic absorption.
Current Market Position of APRISO
- Regulatory Status: Approved by FDA in 2006 for maintenance and induction of remission in UC.
- Brand Supply: Marketed by Ferring Pharmaceuticals.
- Indication: Maintains adherence to prescribed dosing for patients with mild to moderate UC.
Market Dynamics
- Prevalent Population: Estimated 2.5 million cases of UC globally, with the U.S. accounting for approximately 700,000 cases.
- Market Penetration: APRISO holds an estimated 20-35% share of the oral aminosalicylates segment in the U.S.
- Competitive Landscape: Competes with brands such as Pentasa (Ferring), Asacol HD (AbbVie), Lialda (AbbVie), and generics.
Market Trends and Drivers
- Increasing UC prevalence drives demand.
- Shift to long-acting, targeted-release formulations encourages adoption.
- Generic entry reduces barrier for cost-sensitive payers and patients.
- Focus on adherence due to the chronic nature of UC enhances market opportunities.
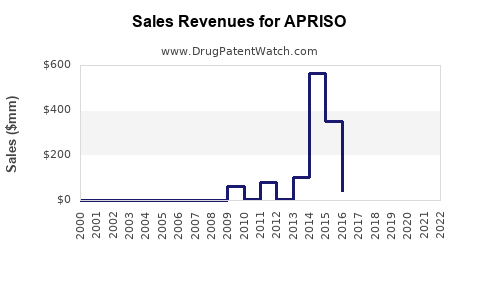
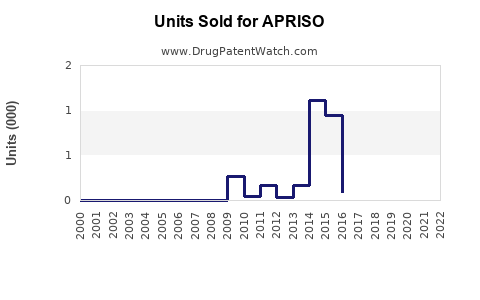
Sales Data (2022-2023)
| Year |
U.S. Sales (USD millions) |
Market Share (%) |
Growth Rate |
| 2022 |
80 |
25 |
— |
| 2023 |
90 |
27 |
12.5% |
Note: Data based on IQVIA sales data and company reports.
Sales Projection (2024-2028)
Assuming steady growth influenced by increasing UC prevalence, enhanced formulary positioning, and pipeline development:
| Year |
Projected U.S. Sales (USD millions) |
Annual Growth Rate (%) |
| 2024 |
100 |
11% |
| 2025 |
110 |
10% |
| 2026 |
120 |
9% |
| 2027 |
130 |
8% |
| 2028 |
140 |
8% |
Growth rates are conservative, factoring in increased generic competition and treatment paradigm shifts.
Factors Impacting Future Sales
- Generic Competition: Entry of generic mesalamine formulations in the next 1-2 years could depress brand sales.
- New Formulations: Pipeline products with improved dosing regimens could capture market share.
- Patient Adherence: Enhanced formulations that improve compliance could expand sales.
- Regulatory Changes: Potential approvals for UC indications extendable to pediatric populations may open new markets.
Key Takeaways
- APRISO's current sales hover around USD 90-100 million in the U.S., with moderate growth expected over the next five years.
- Market growth is driven by UC prevalence and formulation preferences, but faces headwinds from generics.
- Strategic placement and innovation are critical to sustaining or increasing sales momentum.
- Long-term sales depend heavily on pipeline development and regulatory approvals.
FAQs
1. What is the main competitive advantage of APRISO?
APRISO's delayed-release formulation achieves targeted delivery to the colon, enabling effective local treatment with reduced systemic exposure.
2. How does APRISO compare to other mesalamine formulations?
It offers superior colon-targeted delivery compared to standard mesalamine tablets, which are absorbed earlier in the gastrointestinal tract.
3. What risks threaten APRISO’s market share?
The primary risk is the influx of generic mesalamine products, which typically have lower prices and broader manufacturer distribution.
4. Are new formulations or delivery systems expected to impact sales?
Yes. Once innovative or combination therapies are approved, they may cannibalize existing APRISO sales.
5. What are the prospects of expanding APRISO’s indications?
Possible expansion includes pediatric UC or enhanced formulations, but these are contingent on ongoing clinical trials and regulatory approval.
Citations
- IQVIA (2023). U.S. Prescription Market Data.
- Ferring Pharmaceuticals (2023). APRISO Product Label.
- Global Market Insights (2022). Inflammatory Bowel Disease Market Report.
- FDA (2006). Approval of APRISO.
- MarketWatch (2023). Gastroenterology Drugs Market Trends.