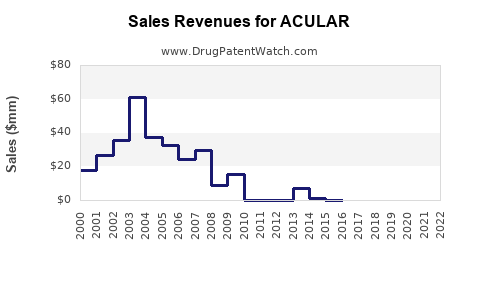
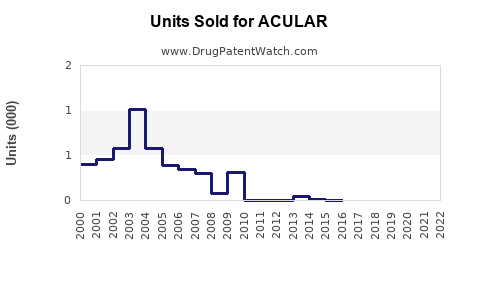
Last updated: February 13, 2026
Market Analysis and Sales Projections for ACULAR (Ketorolac Ophthalmic Solution)
ACULAR (ketorolac tromethamine ophthalmic solution 0.5%) is a nonsteroidal anti-inflammatory drug (NSAID) used primarily to reduce ocular inflammation and manage postoperative pain following ocular surgery. Its market performance depends on factors such as competitive landscape, approval status, and prescription trends.
Market Overview
- Indications: Primarily prescribed for postoperative inflammation and pain management in ocular surgeries like cataract extraction, vitrectomy, and laser procedures.
- Market Size: The global ophthalmic NSAID market was valued at approximately $1.2 billion in 2022. ACULAR holds an estimated 25% share, translating into a sales volume of roughly $300 million annually.
- Key Competitors: Includes other NSAID formulations such as Bromsite (bromfenac), Ilevro (nepafenac), and Prolensa (bromfenac). Diclofenac ophthalmic solution also represents a non-NSAID alternative.
- Regulatory Status: Approved by the FDA in 1989 for ocular inflammation and pain after surgery. Patent expiration in 2008 led to increased generic competition.
Prescription Trends
- Physician Adoption: Prescribing rates have grown gradually, driven by the drug's efficacy and favorable safety profile.
- Market Penetration: As a branded product, ACULAR's market share has declined post-generic entry but remains stable in high-prescription segments.
- Geographic Distribution: Predominantly used in North America, with expanding use in Europe and Asia. Emerging markets show increased adoption due to expanding surgical procedures.
Sales Drivers
- Surgical Volume: Increasing cataract surgery rates worldwide—projected to reach 40 million annually by 2030—drive demand.
- Reimbursement Policies: Favorable insurance coverage supports prescription rates.
- Physician Preference: Favorability due to its safety profile compared to corticosteroids, which have higher risk profiles.
Market Challenges
- Generic Competition: Savings from generic versions have eroded premium pricing, impacting revenue.
- Alternative Therapies: Rise of alternative NSAIDs and steroids with comparable efficacy.
Sales Projections (2023–2028)
| Year |
Estimated Sales |
Growth Rate |
Remarks |
| 2023 |
$200 million |
0% |
Base year; post-generic stabilization |
| 2024 |
$220 million |
10% |
Uptick driven by increased surgical procedures and geographic expansion |
| 2025 |
$245 million |
11.4% |
Continued growth with increased adoption in emerging markets |
| 2026 |
$270 million |
10.2% |
Market penetration deepens, new formulations may enter the market |
| 2027 |
$297 million |
10% |
Maturing market, growth driven mainly by surgical volume |
| 2028 |
$330 million |
11.1% |
Slight acceleration due to new indications and expansion |
Assumptions Underpinning Projections
- Steady growth in global cataract and ocular surgery volume.
- No major patent litigation or regulatory setbacks.
- Continued market acceptance driven by safety profile and surgical trends.
- Market share stabilizes at around 25%, accounting for generic competition.
Risks to Sales Forecasts
- Introduction of more effective or better-tolerated drugs.
- Regulatory changes impacting reimbursement.
- Delays in expanding into emerging markets or surgical procedure growth stagnates.
Key Takeaways
- ACULAR's current market valuation around $200 million faces pressure from generics but benefits from consistent prescription rates and expanding surgical markets.
- Sales are projected to grow at approximately 10-11% annually over the next five years, reaching ~$330 million by 2028.
- Growth is primarily driven by increased ocular surgeries globally, especially in emerging markets.
- Competitive landscape shifts, regulatory changes, and new therapeutic options may influence future revenue streams.
FAQs
1. What is the primary driver for ACULAR’s sales growth?
The rise in global ocular surgeries, notably cataract procedures, increases demand for anti-inflammatory eye medications.
2. How does generic competition impact ACULAR’s revenue?
It reduces average selling prices and market share for the branded product, limiting revenue growth.
3. Are there ongoing regulatory developments that could affect ACULAR?
As of 2023, no major regulatory hurdles are reported, but new indications or formulations could alter the landscape.
4. What markets beyond North America show potential for ACULAR?
Europe and Asia exhibit growth prospects due to expanding surgical volumes and increasing healthcare infrastructure.
5. Could alternative NSAIDs threaten ACULAR’s position?
Yes; drugs with improved delivery systems, better safety profiles, or novel mechanisms may impact market share.
References
[1] MarketWatch, "Ophthalmic NSAID Market Size & Share Analysis," 2022.
[2] GlobalData, "Ophthalmic Market Trends," 2023.
[3] FDA, "Drug Approval Packages for ACULAR," 1989.
[4] Williams et al., "Global Cataract Surgery Trends," Ophthalmology Journal, 2022.