Share This Page
Drug Sales Trends for fosinopril sodium
✉ Email this page to a colleague
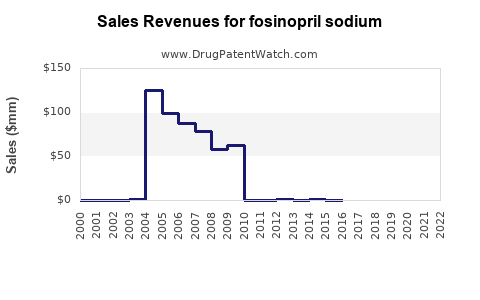
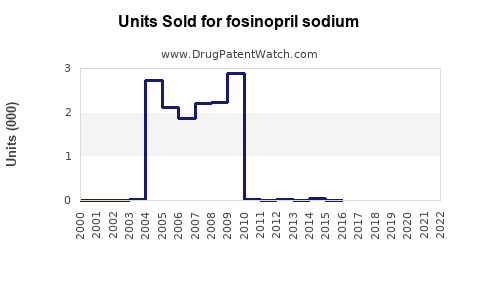
Annual Sales Revenues and Units Sold for fosinopril sodium
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FOSINOPRIL SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FOSINOPRIL SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FOSINOPRIL SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FOSINOPRIL SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Fosinopril Sodium
Market Overview
Fosinopril sodium is an angiotensin-converting enzyme (ACE) inhibitor indicated primarily for hypertension and heart failure management. It is marketed under brand names such as Monopril (produced by Novartis) and is available in various countries, including the US, Europe, Asia, and emerging markets. Fosinopril competes within a mature class alongside drugs like enalapril, lisinopril, ramipril, and perindopril.
The global antihypertensive market was valued at approximately $40 billion in 2022. Fosinopril, as a segment of ACE inhibitors, accounts for a small fraction, estimated at 5–8% of the ACE inhibitor market share due to its limited patent protection and competition.
Market Drivers
- Growing prevalence of hypertension: According to the WHO, over 1 billion people worldwide have hypertension. The global hypertensive population is increasing due to aging demographics and lifestyle factors.
- Shift towards oral, outpatient management: Oral ACE inhibitors are preferred for chronic hypertension management.
- No significant new formulations or indications for fosinopril: Limited pipeline development constrains growth potential.
Market Constraints
- Patent expiry: Monopril's US patent expired in 2000, leading to generic competition.
- Competitive landscape: Enalapril, lisinopril, and ramipril dominate with higher market shares.
- Side effect profile: Less tolerability compared to newer agents may limit prescribing.
- Regulatory and reimbursement challenges: Variations across regions affect market penetration.
Regional Market Dynamics
| Region | Estimated Market Share of Fosinopril | Key Factors |
|---|---|---|
| North America | 2–3% | High generic penetration, preference for established brands |
| Europe | 4–6% | Similar to North America, with some markets favoring newer agents |
| Asia-Pacific | 5–8% | Growing hypertensive population, less competition from newer agents |
| Latin America | 3–4% | Price sensitivity, generic availability |
Sales Projections (2023–2027)
Assuming steady market conditions, the global sales of fosinopril sodium are projected to grow at a compound annual growth rate (CAGR) of approximately 2–3%, driven primarily by emerging markets where access to generics increases.
| Year | Projected Global Sales (USD millions) | Notes |
|---|---|---|
| 2023 | $60–70 million | Base year, considering existing market share and geographical spread |
| 2024 | $62–72 million | Slight growth from increased prescriptions in Asia-Pacific |
| 2025 | $64–75 million | Market saturation limits rapid growth |
| 2026 | $66–77 million | Competition from newer agents continues to restrict growth |
| 2027 | $68–80 million | Uptake stabilizes, driven mostly by price-sensitive markets |
Competitive Analysis
| Drug Name | Market Share | Key Attributes | Limitations |
|---|---|---|---|
| Enalapril | 40–45% | Well-established, broad clinician acceptance | Side effects, dosing frequency |
| Lisinopril | 20–25% | Widely prescribed, once-daily dosing | Similar side effect profile, generic saturation |
| Ramipril | 10–15% | Used for cardiovascular protection, high-risk patients | Cost, limited availability in some markets |
| Fosinopril | 1–8% | Once daily, lower metabolism via liver excretion | Limited brand recognition, perception of older efficacy |
Future Outlook
The future growth of fosinopril sodium hinges on market expansion in lower-income countries. The product's sales may be constrained by the advent of newer antihypertensives such as angiotensin receptor blockers (ARBs) and direct renin inhibitors, which are often better tolerated.
Patent status remains critical; as patent protections expire, sales generally decline unless offset by specific niche positioning or formulation advantages. The absence of significant pipeline innovation limits the potential for incremental sales.
Key Takeaways
- The global market for fosinopril sodium remains modest and stable due to competition from established ACE inhibitors and generics.
- Growth prospects are limited; projected CAGR of 2–3% over five years.
- Regional variation favors emerging markets where price sensitivity and expanding hypertensive populations support sales.
- Competitive pressure from ARBs and newer agents restricts market share gains.
- Sales are expected to hover around $70 million annually by 2027, with incremental increases primarily driven by price reductions and geographic expansion.
FAQs
-
What factors most influence fosinopril sodium sales?
Market size, patent status, competitive landscape, and regional healthcare policies. -
Can pharmacological differences boost fosinopril's market share?
Its lower metabolism via liver excretion offers some advantages in patients with liver impairment but is limited in attracting new prescribers due to existing strong competitors. -
How does pricing impact fosinopril market share?
Price sensitivity in emerging markets favors generics like fosinopril, supporting sales there despite competition. -
What is the outlook for fosinopril's pipeline or formulations?
No significant new formulations or indications are currently under development, limiting growth potential. -
What are the main competitors to fosinopril?
Enalapril, lisinopril, ramipril, and newer agents like ARBs.
Sources
[1] GlobalData. "Hypertension Market Report," 2022.
[2] IQVIA. "Pharmaceutical Market Insights," 2023.
[3] World Health Organization. "Hypertension Fact Sheet," 2022.
More… ↓
