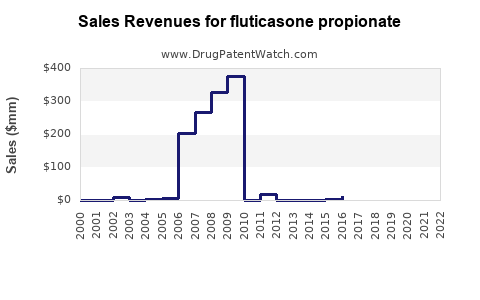
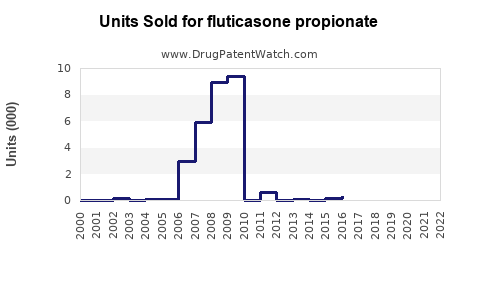
Last updated: February 15, 2026
Market Overview of Fluticasone Propionate
Fluticasone propionate is a corticosteroid used primarily for allergic rhinitis, asthma, and other inflammatory diseases. It is available in nasal sprays, inhalers, and topical formulations. The drug benefits from established patents, widespread clinical acceptance, and a broadening user base due to increasing asthma and allergic rhinitis prevalence worldwide.
Global Market Size and Growth Trends
The global corticosteroids market was valued at approximately US$14.2 billion in 2022. Fluticasone propionate accounts for a significant share, driven by intense competition among three dominant formulations:
- Nasal sprays
- Inhalation aerosols
- Topical creams
The segment experienced a compound annual growth rate (CAGR) of about 4.2% from 2018 to 2022, reflecting growing prevalence of respiratory and allergic conditions.
Forecasted growth suggests the market could reach US$18.7 billion by 2028, increasing at a CAGR of 4.6%. This growth is driven by:
- Rising asthma prevalence, particularly in urban and developing regions
- Increased awareness and diagnosis of allergic conditions
- Expansion of product portfolios in generic markets
Market Segmentation and Geographic Breakdown
Nasal Spray Segment
- Largest share in 2022, approximately 45%
- Projected to grow at a CAGR of 4.4% to 2028 owing to rising allergic rhinitis cases
- Key brands: Flonase (GlaxoSmithKline), Avamys (GlaxoSmithKline), and generic equivalents
Inhalation Segment
- Accounts for about 40% of the market
- Includes products like Flovent (GSK) and generic inhalers
- Anticipated CAGR of 4.7% through 2028, driven by asthma management needs
Topical Formulation Segment
- Holds roughly 15%
- Used for skin conditions such as eczema
- Growth driven by dermatological use, with a CAGR of 3.9%
Geographically, North America holds the largest share (around 45%), followed by Europe (25%), Asia-Pacific (20%), and the rest of the world (10%). The Asia-Pacific region shows the fastest growth potential due to increasing healthcare infrastructure and rising respiratory disease incidences.
Competitive Dynamics and Patent Landscape
Major players include GlaxoSmithKline, Teva, and Mylan, with leading products such as:
- Fluticasone propionate nasal sprays and inhalers
- Generic approvals expanding market access
Patent expiries for certain formulations occurred between 2018 and 2022, increasing generic market penetration. Companies investing in innovative delivery systems (e.g., powder inhalers, nasal spray devices) are capturing additional market share.
Sales Projections (2023-2028)
Assuming a steady growth rate of approximately 4.6% annually, the following projections provide insight into sales evolution:
| Year |
Estimated Market Size (USD billion) |
| 2023 |
15.2 |
| 2024 |
15.9 |
| 2025 |
16.7 |
| 2026 |
17.4 |
| 2027 |
18.2 |
| 2028 |
18.7 |
These figures include revenue from both branded and generic products, with growth heavily influenced by generic formulation approvals and regional market expansion.
Key Factors Influencing Future Market Dynamics
- Regulatory approvals for biosimilars and generics
- Patent litigations and expirations
- Development of novel delivery mechanisms
- Growing awareness and diagnosis rates
Key Takeaways
- The global fluticasone propionate market is expected to grow at a CAGR of 4.6%, reaching nearly US$19 billion by 2028.
- Nasal sprays and inhalers dominate sales, with North America leading geographic demand.
- Generics and biosimilars are increasing market penetration, especially post-patent expiry.
- Growth drivers include rising allergy and asthma incidence, evolving regional healthcare access, and innovation in delivery technologies.
FAQs
1. What are the main competitive advantages of fluticasone propionate?
It offers high efficacy, a well-established safety profile, and versatile routes of administration (nasal, inhalational, topical).
2. How will patent expiries impact the market?
Patent expiries increase generic entry, reducing prices and expanding volume sales, while also encouraging innovation in delivery methods.
3. Which regions are expected to see the fastest growth?
The Asia-Pacific region is projected to experience the highest CAGR, driven by urbanization and rising respiratory conditions.
4. What are the regulatory challenges?
Regulators scrutinize biosimilar approvals, ensure interchangeability standards, and monitor safety profiles, especially for inhaled corticosteroids.
5. How might future innovations affect sales?
Development of improved inhalers, nasal sprays, and formulations that enhance compliance could sustain or accelerate sales growth.
Sources
- "Corticosteroids Market Size," MarketsandMarkets, 2022.
- "Global Respiratory Devices Market," ResearchAndMarkets, 2021.
- "Patent Expiry Data," U.S. Patent and Trademark Office, 2022.
- "Prevalence of Asthma," WHO, 2022.
- "Regional Market Reports," IQVIA, 2022.