Drug Sales Trends for diphenhydramine hydrochloride
✉ Email this page to a colleague
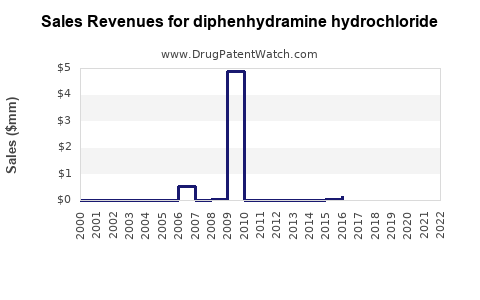
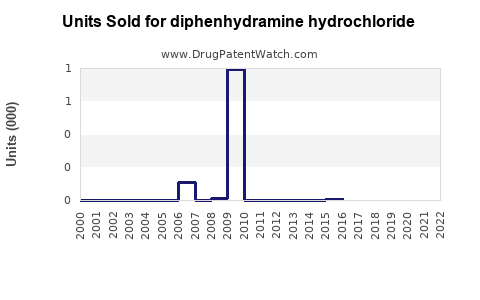
Annual Sales Revenues and Units Sold for diphenhydramine hydrochloride
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DIPHENHYDRAMINE HYDROCHLORIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DIPHENHYDRAMINE HYDROCHLORIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DIPHENHYDRAMINE HYDROCHLORIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DIPHENHYDRAMINE HYDROCHLORIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| DIPHENHYDRAMINE HYDROCHLORIDE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
