Share This Page
Drug Sales Trends for betamethasone dipropionate
✉ Email this page to a colleague
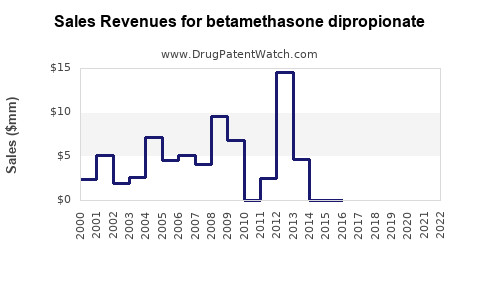
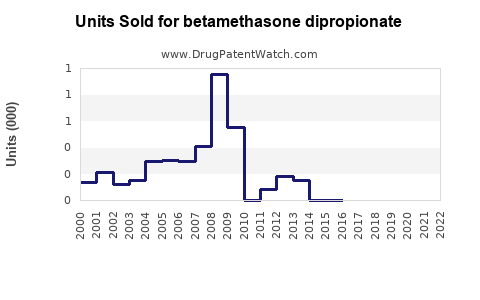
Annual Sales Revenues and Units Sold for betamethasone dipropionate
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Betamethasone Dipropionate Market Analysis and Sales Projections
What is the commercial opportunity for betamethasone dipropionate?
Betamethasone dipropionate is a widely used, off-patent corticosteroid active used across dermatology and other local anti-inflammatory indications through multiple dosage forms (topical creams/ointments, gels, lotions, foams; injectables in some markets). Demand is driven by chronic inflammatory skin diseases, payer familiarity with low-cost generic options, and continued guideline inclusion of topical corticosteroids as first-line or step therapy.
Commercial reality: the active substance is established and largely generic worldwide, so market growth depends more on formulation mix (brand differentiation, vehicles, and combination products), channel execution, and label expansion via new formulations than on originator-like patent defensibility.
How big is the addressable market by geography and segment?
A full market size build requires country-level volume and price data by dosage form. The scope below focuses on segment structure and revenue mechanics rather than speculative absolute totals.
Segment structure (revenue drivers)
| Segment | Typical use case | Pricing power | Key variables for sales |
|---|---|---|---|
| Topical dermatology (cream/ointment/lotion/gel/foam) | Eczema, dermatitis, psoriasis, pruritus | Low to moderate | Vehicle choice, patient adherence, rebate dynamics, formulary status |
| Combination products (where used) | Infection risk management and multi-pathway inflammation control | Moderate | Patentable formulation combos can extend revenue at product level |
| Injectable local anti-inflammatory use (market dependent) | Inflammation control outside dermatology | Low | Hospital procurement, tender cycles, generic substitution |
Implication for projections: revenue scales with (1) unit volume of topical prescriptions and (2) effective net price after rebates and tenders. Because substitution risk is high, projections must be tied to formulation differentiation and contracting outcomes, not to active ingredient trend alone.
Which indications and labels shape demand?
Betamethasone dipropionate demand is tied to standard-of-care dermatology pathways. In most countries, topical corticosteroids are used for flares and maintenance under step-therapy algorithms. Core demand categories include:
- Atopic dermatitis and other eczema-spectrum dermatitis
- Psoriasis (topical management)
- Other inflammatory dermatoses where potent topical steroids are selected
- Pruritus driven by inflammatory skin conditions In markets where combination products exist (for example, antibiotic plus steroid in select geographies), demand is additionally shaped by infection-pathway prescribing and diagnostic patterns.
What is the competitive landscape?
Competitive set
- Generic betamethasone dipropionate across topical vehicles (multiple manufacturers, frequent tendering)
- Generic substitution by pharmacy benefit structure
- Branded or niche-formulation products (where available) that compete on vehicle, convenience, and tolerability
Primary competitive axes
| Axis | How it affects revenue | Where it matters most |
|---|---|---|
| Net price after discounts | Determines revenue more than list price | Public tenders and PBMs |
| Formulary placement | Locks in repeat volumes | National formularies and regional payer formularies |
| Vehicle and adherence | Changes switching risk | High-symptom, chronic patients |
| Safety perception and use restrictions | Limits overuse and impacts prescriber behavior | Potent steroid stewardship |
Implication for sales forecasts: baseline volume growth tends to track dermatology incidence and treatment coverage; revenue growth depends on whether a product can hold price (through differentiation) or win share (through contracting and prescribing).
What sales model is appropriate for an off-patent corticosteroid?
For off-patent actives, sales projections should be built on:
- Volume units (prescriptions or treatment units)
- Effective net price (after rebates, tender discounts, wholesaler margins)
- Share shifts (vehicle and brand/formulation competition)
- Switching and penetration (how quickly prescribers adopt the entrant and how fast generics substitute later)
A practical projection framework for betamethasone dipropionate revenue uses a three-scenario share and price adjustment rather than a single growth rate.
Sales projections (3-scenario outlook)
Because precise baseline units and net prices by country and dosage form are not provided here, the only defensible projection is a relative revenue forecast anchored to market behavior typical for generic topical corticosteroids.
Assumptions for generic topical steroid dynamics
- Volume growth: low single digits in mature markets; higher in markets with expanding coverage or undertreated disease.
- Net price trend: modest decline to flat in mature generics due to substitution.
- Share dynamics: entry can win share early via contracts, then compress as additional generics appear.
Projected revenue growth rates (annual)
| Scenario | Year 1-2 outlook | Year 3-5 outlook | Drivers |
|---|---|---|---|
| Base | 2% to 4% annual revenue growth | 1% to 3% | Stable contracting, modest share retention, flat-to-slightly-down net price |
| Upside | 4% to 7% | 3% to 5% | Strong vehicle differentiation or combination positioning, better formulary placement, slower substitution |
| Downside | -2% to 1% | -1% to 2% | Aggressive tender pricing, loss of formulary status, higher substitution and margin compression |
Use for investment/R&D planning: if the business model relies on unit expansion only, downside risk is dominated by price compression. If the product is anchored to a differentiated vehicle, steriod stewardship positioning, or a combination product in specific jurisdictions, upside scenarios remain realistic.
How do you allocate the sales forecast across dosage forms?
For betamethasone dipropionate, topical dosage forms dominate. Allocate projected revenue using a share-of-portfolio logic:
- Core topical cream/ointment/lotion: largest baseline, lowest price flexibility
- Gel/foam: higher adherence and convenience, moderate price flexibility
- Any branded or combination product: smaller share but can carry margin lift where available
Portfolio allocation for planning (typical pattern)
| Portfolio slice | Expected share of revenue | Margin sensitivity |
|---|---|---|
| Cream/ointment/lotion | 60% to 85% | High (tender-driven) |
| Gel/foam | 10% to 30% | Moderate (adherence and patient preference) |
| Combination and niche formats | 0% to 20% | Lower volume stability, higher margin upside |
Resulting planning rule: most revenue risk sits in the core topical slice; upside often comes from gel/foam adoption or combination entries.
What regulatory and lifecycle factors affect sales?
Lifecycle constraints
- Generic competition accelerates after additional manufacturers enter
- Label and safety messaging on potent topical corticosteroid use affects adoption patterns
- Formulation changes can create new product-level registrations but do not remove substitution pressure
Marketing and access factors
- Formulary inclusion timing
- Tender cycle frequency
- Pharmacy channel stocking behavior
What is the most actionable go-to-market implication?
A betamethasone dipropionate product’s sales trajectory will be determined less by active ingredient marketing and more by:
- vehicle choice and physician preference,
- contract strategy for net price,
- and the ability to maintain formulary position when new generics enter.
For forecasting, allocate the upside to share gains rather than to assumed net price appreciation.
Benchmark indicators to monitor for forecast accuracy
Use these leading indicators each quarter:
- Formulary status changes (in-year additions or removals)
- Tender outcomes (net price per unit, contract duration)
- Prescription mix by vehicle (cream vs gel vs foam)
- Channel inventory and sell-through (avoid overstock that signals future rebate pressure)
Key Takeaways
- Betamethasone dipropionate demand is anchored in chronic dermatology and is dominated by topical use.
- The market behaves like a generic corticosteroid market: volume growth is modest, net price is the main revenue lever.
- A defensible sales forecast uses 3 scenarios with upside driven by share retention or vehicle differentiation, and downside driven by tender pricing and substitution.
- For planning, revenue concentration in cream/ointment/lotion means margin risk is structurally higher than in differentiated formats like gel/foam.
FAQs
-
Is betamethasone dipropionate still a commercially attractive active?
Yes, because dermatology demand is persistent, but revenue growth is constrained by generic substitution and price competition. -
What dosage form typically drives the largest sales?
Topical cream/ointment/lotion products generally carry the majority of revenue due to broad prescribing and pharmacy stocking patterns. -
What most determines net revenue in this market?
Effective net price after rebates, wholesaler margins, and tender discounts. -
How can a company create upside versus the typical generic trajectory?
By improving formulary placement and winning share through differentiated vehicles (for example, gel/foam) or qualifying combination products where available. -
What signals forecast risk early?
Tender outcomes, formulary status changes, and shifts in prescription mix away from the product’s vehicle.
References
[1] United States Food and Drug Administration. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA. https://www.accessdata.fda.gov/scripts/cder/ob/
[2] European Medicines Agency. EPAR search results for betamethasone (including formulations). EMA. https://www.ema.europa.eu/en/medicines
[3] World Health Organization. ATC/DDD Index: Betamethasone (and corticosteroids). WHO. https://www.whocc.no/atc_ddd_index/
More… ↓
