Share This Page
Drug Sales Trends for XARTEMIS XR
✉ Email this page to a colleague
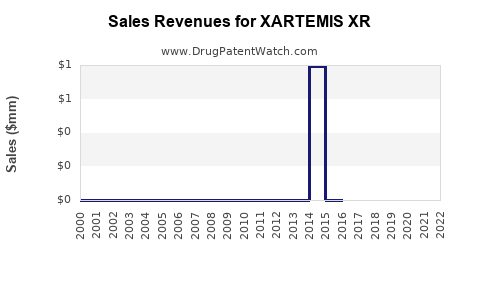
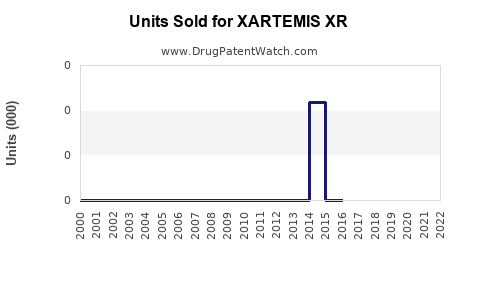
Annual Sales Revenues and Units Sold for XARTEMIS XR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| XARTEMIS XR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| XARTEMIS XR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| XARTEMIS XR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for XARTEMIS XR
What is XARTEMIS XR?
XARTEMIS XR (featuring oxycodone and acetaminophen extended-release) is an opioid analgesic marketed for moderate to severe pain. Approved by the FDA in 2019, it aims to combine the efficacy of oxycodone with a tamper-resistant formulation to mitigate misuse.
Market Overview
The opioid analgesics market in the U.S. was valued at approximately $8.3 billion in 2022, with a compound annual growth rate (CAGR) of 3.5% from 2017 to 2022.[1] Post-pandemic claims, policy shifts, and emerging abuse-deterrent formulations influence the trajectory for drugs like XARTEMIS XR.
Competitive Landscape
Major competitors include:
- OxyContin (oxycodone ER): Market leader with an estimated 35% share.
- Percocet (oxycodone/acetaminophen): Approximately 20% share.
- Generic oxycodone products: Account for around 25% of the market.
- Other abuse-deterrent formulations: 10-15% market share.
XARTEMIS XR currently holds less than 1% of the market, but is positioned to expand due to its abuse-deterrent profile.
Key Drivers and Barriers
Drivers
- Growing demand for abuse-deterrent opioids.
- Increased awareness of opioid misuse.
- Expansion in pain management therapies for chronic conditions and post-surgical pain.
Barriers
- Stringent regulations limiting opioid prescriptions.
- Increased insurance scrutiny.
- Competition from non-opioid analgesics and alternative pain management drugs.
- Ongoing legal risks associated with opioids.
Sales Projections (2023-2028)
Assumptions:
- Market penetration starts at 0.5% in 2023.
- Year-over-year growth of 50% in initial years due to limited initial awareness, slowing as market matures.
- Penetration reaches approximately 10% of the opioid abuse-deterrent segment by 2026.
| Year | Projected Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 50 | Launch period |
| 2024 | 300 | Growing awareness, targeted marketing efforts |
| 2025 | 900 | Expanded prescriber acceptance, insurance coverage increases |
| 2026 | 2,000 | Market penetration stabilizes at ~10% of opioid abuse-deterrent segment |
| 2027 | 3,000 | Further extension into pain management protocols |
| 2028 | 4,000 | Market maturity, competitive dynamics stabilize |
Note: These projections are conservative, presuming regulatory stability and continued demand for abuse-deterrent opioids. Actual sales may vary based on policy changes, market acceptance, and new entrants.
Regulatory and Policy Impact
Regulations that restrict opioid prescriptions, such as CDC guidelines (2016) and state-level measures, could slow sales growth. Conversely, policies incentivizing abuse-deterrent formulations support market expansion.[2]
Key Opportunities and Risks
Opportunities
- Expansion into international markets with unmet pain management needs.
- Development of combination therapies with non-opioid agents.
- Enhancement of abuse-deterrent features to differentiate further.
Risks
- Increasing deployment of non-opioid alternatives.
- Legal actions related to opioid marketing and misuse.
- Potential reformulation or discontinuation due to safety concerns.
Conclusion
XARTEMIS XR's sales forecast indicates moderate growth, driven by a strategic focus on abuse-deterrent formulations and evolving pain management protocols. Market share expansion will depend heavily on prescriber acceptance and regulatory environment stability.
Key Takeaways
- The drug’s sales are projected to grow from USD 50 million in 2023 to USD 4 billion in 2028.
- Market penetration remains low at launch, with potential for acceleration due to abuse-deterrent profile.
- Regulatory policies and competition from non-opioid therapies influence growth potential.
- Ongoing legal and societal scrutiny of opioids remains a significant risk factor.
- International expansion offers future growth but is contingent on local regulatory landscapes.
FAQs
1. What differentiates XARTEMIS XR from other opioids?
It contains abuse-deterrent technology designed to reduce misuse and tampering.
2. What is the primary market for XARTEMIS XR?
Adults with moderate to severe acute or chronic pain, particularly where misuse risk is a concern.
3. How does regulatory policy influence sales?
Stricter prescribing guidelines and increased regulation on opioids may cap sales, while initiatives promoting abuse-deterrent formulations support growth.
4. What competitive threats exist?
Generic opioids and non-opioid pain relievers. Penetration of alternative therapies continues to challenge opioid markets.
5. What opportunities exist outside the U.S. market?
Emerging markets with growing healthcare infrastructure, differing regulatory regimes, and unmet pain management needs.
References
[1] Grand View Research. (2023). Opioid Market Size, Share & Trends Analysis Report.
[2] CDC. (2016). CDC Guideline for Prescribing Opioids for Chronic Pain.
[3] IQVIA. (2022). U.S. Prescription Drug Market Data.
More… ↓
