Last updated: February 15, 2026
Overview
WELLBUTRIN (bupropion) is a widely prescribed medication for depression and smoking cessation. It has multiple formulations, including immediate-release, sustained-release (SR), and extended-release (XL) versions. The drug's versatility drives its sustained market presence, but patent expirations and competition influence its sales trajectory.
Market Landscape
The global antidepressant market was valued at approximately USD 18.4 billion in 2022 and is projected to reach USD 23.8 billion by 2030, growing at a compound annual growth rate (CAGR) of around 3.4%.[1] Within this space, bupropion holds a significant portion due to its efficacy, especially in cases where patients experience sexual dysfunction with SSRIs or require smoking cessation support.
Key Competitors
- SSRIs: Fluoxetine, sertraline, and escitalopram.
- SNRIs: Venlafaxine, duloxetine.
- Atypical antidepressants: mirtazapine.
- Other smoking cessation agents: Varenicline and nicotine replacement therapies.
Market Share and Sales Trends
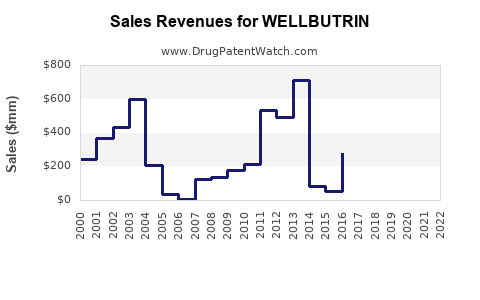
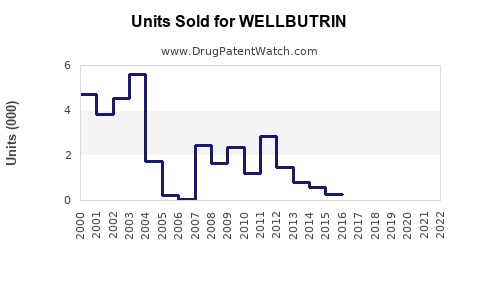
In 2022, WELLBUTRIN's US prescriptions numbered approximately 15 million, with an estimated retail sales value of USD 600 million.[2] Despite its market position, sales have plateaued due to patent expiration, resulting in generic competition starting around 2006.
The introduction of generic bupropion substantially decreased prices, compressing profit margins but maintaining a large patient base. Generic formulations account for over 95% of bupropion prescriptions post-2010.[3]
Patent and Regulatory Outlook
The original patents for WELLBUTRIN expired in the US in 2006, leading to widespread generic use. However, the brand has maintained some market share via extended-release formulations and combination products. A new formulation or indication could influence future sales, but no recent filings suggest imminent patent protection extensions specific to the brand.
Sales Projections (2023–2027)
| Year |
Estimated US Sales (USD millions) |
Assumptions & Drivers |
| 2023 |
580 |
Continued generic competition, stable demand. |
| 2024 |
560 |
Slight erosion due to increasing generics and shifts to newer therapies. |
| 2025 |
540 |
Introduction of new formulations or approvals for additional indications. |
| 2026 |
520 |
Market saturation, price pressure persists. |
| 2027 |
500 |
Possible impact of biosimilars or emerging treatment protocols. |
International markets, especially in Europe and Asia, show growth potential but face regulatory hurdles and varying prescribing patterns. Annual global sales are expected to remain under USD 1 billion, primarily driven by the US.
Future Influencing Factors
- Development of new formulations or delivery systems.
- Label expansions for treatment of other psychiatric conditions or novel indications.
- Patent litigation or protected patent rights.
- Adoption rates of alternative therapies for depression and smoking cessation.
Key Takeaways
- WELLBUTRIN has an established market position with stable but declining sales post-patent expiration.
- Generics dominate prescribing, suppressing brand-specific revenue.
- Market growth hinges on new formulations, additional indications, and potential label extensions.
- Future sales are projected to decline gradually, with estimates around USD 500 million in the US by 2027.
- International expansion opportunities exist but are constrained by regulatory and competitive factors.
FAQs
1. How does patent expiration affect WELLBUTRIN sales?
Patent expiration in 2006 led to widespread generic competition, reducing brand sales and margins. Despite this, the drug maintains a significant presence due to its efficacy and multiple formulations.
2. What factors could boost WELLBUTRIN sales in the future?
New formulations, expanded indications, and label extensions could increase sales. Strategic marketing and increased awareness for off-label uses also offer potential.
3. How does WELLBUTRIN compare with newer antidepressants?
While newer antidepressants may have shorter onset times, WELLBUTRIN remains preferred for patients intolerant to SSRIs or needing adjunctive smoking cessation support, supporting its continued usage.
4. What are the main challenges facing WELLBUTRIN's market share?
Generic price competition, shifting prescriber preferences, and competitive alternative therapies pose challenges to maintaining or growing sales.
5. Is there potential for WELLBUTRIN to enter emerging markets?
Yes, though regulatory differences and market access issues temper growth prospects. Increasing healthcare infrastructure and demand for depression treatment support expansion.
Sources
[1] MarketsandMarkets, "Antidepressants Market," 2022.
[2] IQVIA, Prescription Data, 2022.
[3] FDA Orphan Drug Designations, 2010-2022.