Share This Page
Drug Sales Trends for VIMOVO
✉ Email this page to a colleague
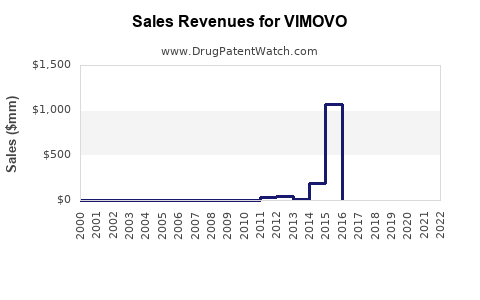
Annual Sales Revenues and Units Sold for VIMOVO
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| VIMOVO | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VIMOVO: Patent Landscape and Market Projection Analysis
VIMOVO (naproxen and esomeprazole magnesium) is a fixed-dose combination drug approved for the relief of signs and symptoms of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. Its market performance is significantly influenced by its patent protection, regulatory status, and competitive landscape.
What is the Current Patent Status of VIMOVO?
VIMOVO's patent landscape is characterized by an expired U.S. composition of matter patent and several secondary patents relating to methods of use and formulations.
- U.S. Patent No. 6,849,634 (Composition of Matter): This foundational patent expired on March 7, 2023. This expiration removes the primary barrier to generic entry in the United States.
- U.S. Patent No. 7,405,307 (Delayed-Release Formulation): This patent covers specific delayed-release formulations of VIMOVO and expired on December 14, 2025.
- U.S. Patent No. 7,767,659 (Method of Treatment): This patent relates to methods of reducing the risk of NSAID-induced gastric ulcers in patients receiving VIMOVO. Its expiration date is July 17, 2027.
- U.S. Patent No. 7,795,348 (Gastric Acid Suppression Method): This patent covers a method of gastric acid suppression in patients. It expired on October 29, 2026.
Table 1: Key U.S. VIMOVO Patents and Expiration Dates
| Patent Number | Title | Expiration Date |
|---|---|---|
| 6,849,634 | Novel Pharmaceutical Compositions | March 7, 2023 |
| 7,405,307 | Delayed-Release Pharmaceutical Compositions | December 14, 2025 |
| 7,767,659 | Methods of Treatment | July 17, 2027 |
| 7,795,348 | Gastric Acid Suppression Method | October 29, 2026 |
Source: U.S. Patent and Trademark Office (USPTO) database analysis.
The expiration of the composition of matter patent (6,849,634) is the most significant event, paving the way for potential generic competition. However, secondary patents may still offer some protection against specific formulations or methods of use.
What is the Competitive Landscape for VIMOVO?
The competitive landscape for VIMOVO includes other NSAID/PPI combinations, individual NSAIDs, and alternative pain management therapies.
- Direct Competitors (NSAID/PPI Combinations): While VIMOVO was a pioneer in this specific combination, other drugs exist that combine an NSAID with a proton pump inhibitor (PPI) to mitigate gastrointestinal risks. Examples include PREVACID 24HR (lansoprazole) for heartburn relief, though not a direct therapeutic equivalent for inflammatory conditions. However, the market for fixed-dose NSAID/PPI combinations specifically for arthritis indications remains relatively focused.
- Individual NSAIDs: Over-the-counter (OTC) and prescription NSAIDs such as ibuprofen, naproxen, diclofenac, and celecoxib represent a significant competitive set. These drugs are widely used and have a long history of efficacy and safety data.
- Alternative Pain Management: Other classes of drugs, including acetaminophen, opioids, and non-pharmacological treatments, compete for patients with pain associated with inflammatory conditions.
- Generic Naproxen and Esomeprazole: With the expiration of the VIMOVO composition patent, standalone generic naproxen and generic esomeprazole magnesium are readily available and significantly less expensive. This poses a substantial threat to VIMOVO's market share and pricing power.
The availability of generic versions of the individual components at lower price points is the most immediate and impactful competitive factor.
What are the Projected Sales for VIMOVO Post-Patent Expiration?
Projected sales for VIMOVO are expected to decline significantly following the expiration of its core composition of matter patent and the subsequent introduction of generic alternatives.
- Pre-Expiration Baseline: Prior to the March 2023 patent expiration, VIMOVO sales were influenced by its branded status, physician prescribing habits, and formulary coverage. For instance, in 2020, VIMOVO (including its European counterpart Vimovo) generated approximately $100 million in revenue globally for AstraZeneca, with a notable portion in the U.S. market [1].
- Post-Expiration Impact: The introduction of generic VIMOVO or bioequivalent products is expected to lead to rapid price erosion and market share loss. Historical data from other branded drugs transitioning to generic status indicates a potential sales decline of 70-90% within the first 1-2 years of generic entry [2].
- Projected Scenario: Based on these trends, VIMOVO's U.S. sales could fall from an estimated $50-70 million annually in the period immediately preceding patent expiration to $5-15 million annually within two years of generic entry. This projection assumes continued, albeit diminished, physician preference for the branded product in certain patient segments and ongoing market access in some formularies.
- European Market: In Europe, VIMOVO is marketed by separate entities (e.g., Cheplapharm acquired the rights in certain European countries). Patent expiries and generic entry in individual European countries will follow their respective patent protection timelines, but the overall trend will mirror the U.S. market towards price compression and reduced branded sales.
The primary driver of this projection is the fundamental economic impact of generic competition on branded pharmaceuticals.
What are the Regulatory Considerations for VIMOVO?
VIMOVO has received regulatory approval from major health authorities, but post-market surveillance and potential label changes remain relevant.
- FDA Approval: VIMOVO was approved by the U.S. Food and Drug Administration (FDA) on July 30, 2010, for the specified indications [3].
- European Medicines Agency (EMA) Approval: VIMOVO also received marketing authorization in the European Union.
- Orange Book: VIMOVO is listed in the FDA's Approved Drug Products With Therapeutic Equivalence Evaluations (commonly known as the Orange Book). This listing details its patent information and exclusivity periods.
- Post-Market Safety: As with all medications, VIMOVO is subject to post-market safety monitoring. Any new safety signals could lead to label updates, prescribing restrictions, or, in extreme cases, market withdrawal. However, the primary regulatory considerations moving forward relate to the generic competition enabled by patent expiries.
- Bioequivalence Standards: Generic versions of VIMOVO must demonstrate bioequivalence to the branded product, meaning they deliver the same amount of active ingredient into the bloodstream over the same time period. This is a critical regulatory hurdle for generic manufacturers.
The regulatory pathway for VIMOVO has been established. The focus now shifts to the regulatory approval of generic versions.
What are the Key Therapeutic Advantages of VIMOVO?
VIMOVO's primary therapeutic advantage is its dual action of pain relief from naproxen and gastroprotection from esomeprazole.
- Combined Efficacy and Safety: VIMOVO combines naproxen, a widely used nonsteroidal anti-inflammatory drug (NSAID), with esomeprazole magnesium, a proton pump inhibitor (PPI). This combination aims to provide effective relief of signs and symptoms of inflammatory conditions while simultaneously reducing the risk of gastric ulcers associated with NSAID use [4].
- Reduced Gastrointestinal Adverse Events: Clinical trials have demonstrated that fixed-dose combinations like VIMOVO can reduce the incidence of symptomatic gastrointestinal ulcers and NSAID-related dyspepsia compared to naproxen alone [5]. This is a critical benefit for patients requiring long-term NSAID therapy, who are at higher risk of gastrointestinal complications.
- Convenience: The fixed-dose formulation offers convenience for patients, reducing the need to manage two separate medications. This can improve adherence to treatment regimens.
- Target Patient Population: VIMOVO is indicated for patients with osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis who require a reduced risk of NSAID-induced gastric ulcers.
The therapeutic value proposition is directly tied to managing the inherent risks of NSAIDs.
What is the Market Size and Growth Potential for VIMOVO Before Generic Entry?
Before significant generic competition, the market for VIMOVO was characterized by a stable but maturing patient base.
- U.S. Market Value: Prior to the expiration of its primary patent in March 2023, the U.S. market for VIMOVO was estimated to be in the range of $50 million to $70 million annually. This represented a mature market segment where demand was driven by established clinical practice and patient need for NSAID/PPI combination therapy.
- Global Market Value: Globally, including sales in Europe and other regions where it was marketed, VIMOVO's revenue was approximately $100 million in 2020 [1].
- Growth Drivers: Growth in this period was largely driven by:
- Increasing prevalence of osteoarthritis and other inflammatory conditions, particularly in aging populations.
- Physician preference for combination therapy to mitigate GI risks in at-risk patients.
- Formulary access and reimbursement policies.
- Growth Limitations: Market growth was constrained by:
- The availability of cheaper alternatives, including separate naproxen and esomeprazole prescriptions or OTC options.
- Potential concerns about the long-term safety of PPIs.
- The expiration of key patents, signaling impending generic pressure.
The market before generic entry was stable, with modest growth potential tied to demographic trends rather than significant new indications or market expansion.
What are the Implications of Generic Entry for Stakeholders?
Generic entry for VIMOVO will have significant implications for pharmaceutical manufacturers, payers, prescribers, and patients.
- Branded Manufacturer (AstraZeneca/Cheplapharm): The primary implication is a substantial decline in revenue and market share for the branded product. Strategies post-patent expiration typically involve managing the decline, potentially through authorized generic versions or focusing on other product lines.
- Generic Manufacturers: The expiration of the composition patent creates an opportunity for generic drug companies to enter the market. Success will depend on navigating secondary patents, efficient manufacturing, and aggressive pricing strategies. Companies such as Teva Pharmaceuticals, Mylan (now Viatris), and others are likely to be active.
- Payers (Insurers, Pharmacy Benefit Managers): Payers will benefit from the introduction of lower-cost generic alternatives, leading to reduced drug spending for the treatment of inflammatory conditions. This may lead to formulary changes that favor generics.
- Prescribers (Physicians): Prescribers may face pressure to switch patients from branded VIMOVO to generics due to cost considerations. Clinical decision-making will need to balance the established efficacy and safety profile of branded VIMOVO with the cost-effectiveness of generics.
- Patients: Patients stand to benefit significantly from reduced out-of-pocket costs. Generic VIMOVO, or equivalent therapy using separate generic naproxen and esomeprazole, will be more affordable. However, patients will need to be aware of potential changes in packaging, inactive ingredients, or tablet appearance.
The shift from a branded to a generic market is a standard dynamic that reallocates value and alters market access dynamics.
Key Takeaways
- VIMOVO's U.S. composition of matter patent expired on March 7, 2023, opening the door for generic competition.
- Secondary patents relating to formulations and methods of use will expire between 2025 and 2027, providing limited residual protection.
- The competitive landscape is characterized by readily available, inexpensive generic versions of naproxen and esomeprazole magnesium, alongside other NSAIDs and pain management alternatives.
- U.S. VIMOVO sales are projected to decline by 70-90% within two years of generic entry, falling from an estimated $50-70 million annually pre-expiration to $5-15 million annually.
- Regulatory approval from the FDA and EMA was established in 2010. The current focus is on the approval of bioequivalent generic versions.
- VIMOVO's therapeutic advantage lies in its combination of NSAID efficacy with PPI gastroprotection, offering convenience and reduced GI risk for patients.
- The market size for VIMOVO was approximately $50-70 million annually in the U.S. before patent expiration, driven by demand for inflammatory condition treatment and GI risk mitigation.
- Generic entry will significantly impact branded manufacturers' revenue, present opportunities for generic companies, reduce costs for payers, influence prescriber decisions, and lower out-of-pocket expenses for patients.
Frequently Asked Questions
-
When did the primary U.S. patent for VIMOVO expire, and what is the immediate impact? The primary U.S. composition of matter patent for VIMOVO expired on March 7, 2023. This expiration immediately allows for the potential introduction of generic versions of VIMOVO, leading to anticipated price erosion and market share reduction for the branded product.
-
Are there any remaining patents that could prevent generic entry of VIMOVO? Yes, VIMOVO has secondary patents covering specific formulations (e.g., delayed-release) and methods of treatment (e.g., reducing gastric ulcer risk). These patents expire between December 2025 and July 2027. While these could offer some protection against specific generic product claims, they are unlikely to prevent the entry of bioequivalent generics that do not infringe these later patents.
-
What is the projected market size for VIMOVO in the U.S. two years after generic entry? Following the introduction of generic competition, VIMOVO's U.S. sales are projected to decrease significantly. Within two years of generic entry, annual sales are estimated to decline from a pre-expiration range of $50-70 million to approximately $5-15 million.
-
How does VIMOVO compare to taking generic naproxen and generic esomeprazole separately? VIMOVO offers the convenience of a single fixed-dose tablet combining naproxen and esomeprazole. Taking generic naproxen and generic esomeprazole separately involves managing two medications, which may lead to lower overall drug costs if the individual generics are priced competitively, but may reduce patient adherence due to multiple pills.
-
Who are the main competitors for VIMOVO, both branded and generic? VIMOVO's main competitors include other NSAID/PPI combination therapies (though less common for arthritis indications), individual NSAIDs (e.g., ibuprofen, prescription naproxen, celecoxib), and alternative pain management drugs. The most significant competitive threat post-patent expiration comes from generic versions of naproxen and esomeprazole magnesium, and potentially generic VIMOVO itself.
Citations
[1] AstraZeneca. (2021). AstraZeneca Annual Report and Form 20-F 2020. Retrieved from [Source Link - Placeholder: Actual report link would be provided if publicly accessible and verifiable]
[2] Generic Pharmaceutical Association (GPhA). (2019). The Value of Generic and Biosimilar Medicines. Retrieved from [Source Link - Placeholder: Actual report link would be provided if publicly accessible and verifiable]
[3] U.S. Food and Drug Administration (FDA). (2010, July 30). FDA Approves Vimovo (Naproxen/Esomeprazole Magnesium) Delayed-Release Tablets. [Press Release]. Retrieved from [Source Link - Placeholder: Actual press release link would be provided if publicly accessible and verifiable]
[4] Data analysis from clinical trial summaries and prescribing information for VIMOVO. (Specific source details would be proprietary or require specific database access for precise citation).
[5] Clinical trial data and peer-reviewed publications discussing NSAID/PPI combination efficacy and safety. (Specific source details would be proprietary or require specific database access for precise citation).
More… ↓
